Heat of Reaction
The heat of reaction, also known as enthalpy of reaction, is the enthalpy change of a chemical reaction occurring at a constant pressure. Generally, it is determined as the energy required to release or produce one mole of a substance [1-4].
Formula [1-4]
Consider the following reaction.
aA + bB → cC + dD
The heat of reaction (ΔHrxn) is given by
ΔHrxn = [c ΔHfo(C) + d ΔHfo(D)] – [a ΔHfo(A) + b ΔHfo(B)]
The general expression for the heat of reaction is given by the difference between the sums of the enthalpies of formation of the products and the reactants.
ΔHrxn = ΣnΔHfo (products) – ΣmΔHfo (reactants)
Where
n: Stoichiometric coefficients of the products
m: Stoichiometric coefficients of the reactants
Symbol: ΔHrxn
Unit: kJ/mol
Characteristics
- ΔHrxn can be positive or negative.
- A positive value indicates that the enthalpy of products is greater than that of the reactants. In other words, the reaction is endothermic and requires heat.
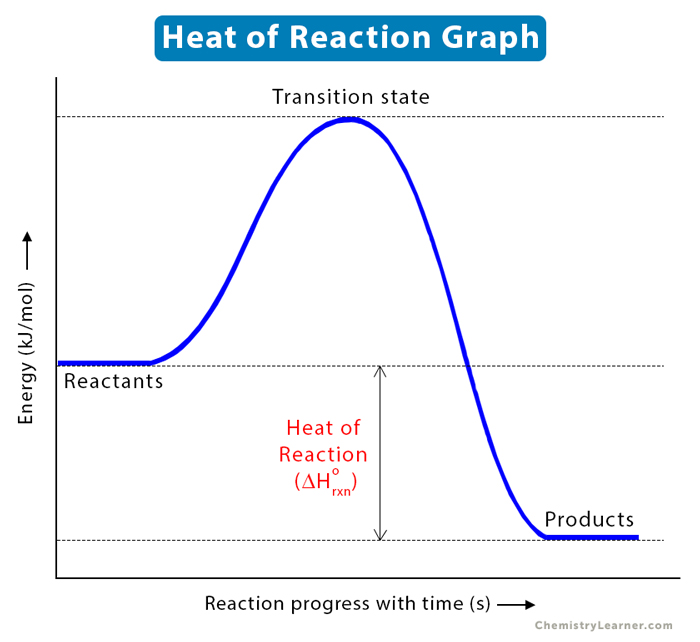
- A negative value indicates that the enthalpy of products is less than that of the reactants (see graph below). In other words, the reaction is exothermic and liberates heat.
- When the reaction occurs at standard temperature and pressure, the heat of reaction is denoted by the symbol ΔHrxno. Standard temperature and pressure mean temperature of – 25 ˚C and pressure of 1 atm.
Examples
1. Let us take the example of the reaction between carbon (C) and oxygen (O2) to form carbon dioxide (CO2) [1-4].
C (s) + O2 (g) → CO2 (g)
The enthalpy of reaction is given by
ΔHrxno = (1 mol) ΔHfo{CO2(g)} – [(1 mol) ΔHfo{C(s)} + (1 mol) ΔHfo{O2(g)}]
Because both C(s) and O2(g) are in their natural and stable form, their enthalpies of formation are zero. Also, the standard enthalpy of formation values can be found in our other article.
Therefore,
ΔHrxno = (1 mol) (-392.505 kJ/mol) – [(1 mol) (0) + (1 mol) (0)}
=> ΔHrxno (CO2) = -392.505 kJ/mol
2. Consider the reaction between methane (CH4) and oxygen (O2) to produce carbon dioxide (CO2) and water (H2O).
CH4 (g) + 2 O2 (g) → CO2 (g) + 2 H2O (l)
ΔHrxno = (1 mol) ΔHfo{CO2(g)} + (2 mol) ΔHfo{H2O (l)} – [(1 mol) ΔHfo{CH4(g)} + (2 mol) ΔHfo{O2(g)}]
=> ΔHrxno = (1 mol) (- 393.5 kJ/mol) + (2 mol) (- 285.8 kJ/mol) – [(1 mol) (-74.81 kJ/mol) + (2 mol) (0)]
=> ΔHrxno = -890.29 kJ/mol
How to Find Heat of Reaction Experimentally
The heat of reaction is usually determined by measuring the heat produced over time using a reaction calorimeter, such as a heat flow calorimeter. A calorimeter is an isolated system that maintains constant pressure. The following equation gives the amount of heat produced [1-4].
q = m cP ΔT
Where,
q: Heat
m: Mass of the reactants
cp: Specific heat of the reactants
ΔT: Change in temperature
The value of q is numerically equal to ΔHrxn, provided we scale it to per mole. The process of measuring changes in enthalpy in this way is called calorimetry.
Example Problem
Calculate the heat of reaction when a given quantity of ethanol is burnt in air to increase the temperature of 400 g of water from 26 to 44 ˚C. The specific heat of water is 4.2 J/g.K.
Solution:
Given, m = 400 g cP = 4.2 J/g.K ΔT = 44 ˚C – 26 ˚C = 18 ˚C
The amount of heat lost in the combustion process equals the heat gained by the water. Therefore, we use the following equation:
q = m cP ΔT
q = 400 g x 4.2 J/g.˚C x 18 ˚C
q = 30240 J or 30.24 KJ
The heat of reaction of ethanol is – 30.24 kJ.