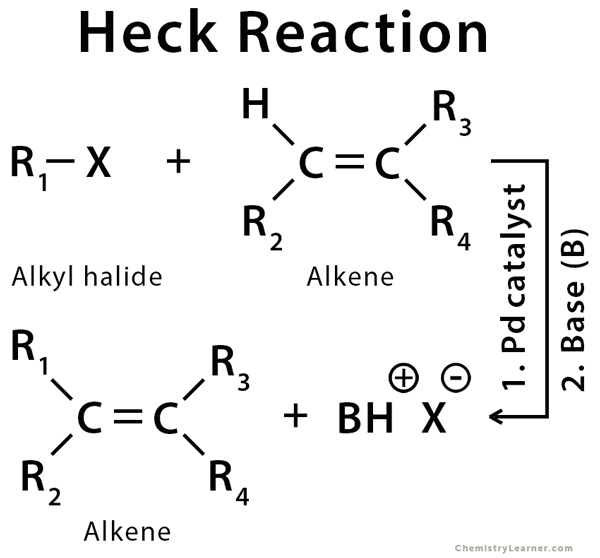
Heck Reaction
Definition: What is Heck Reaction?
A cross-coupling reaction in organic chemistry is a reaction where two fragments are joined together (eg., C-C bonding) with the aid of a metal catalyst. The Heck reaction (also called Mizoroki-Heck reaction) is a cross-coupling reaction of an organohalide with an alkene to make a substituted alkene using palladium as a catalyst and a base. It has drawn much attention due to high efficiency and simplicity. The Heck reaction is significant from a synthetic point of view because of its high chemoselectivity and mild reaction conditions. It has low toxicity and lower cost of the reagent because the catalyst can be recycled [1-3].
The history of this reaction goes back to 1972 when American chemist Richard Heck discovered it. Japanese chemist Tsutomu Mizoroki developed a more practical version of Heck’s initial reaction.
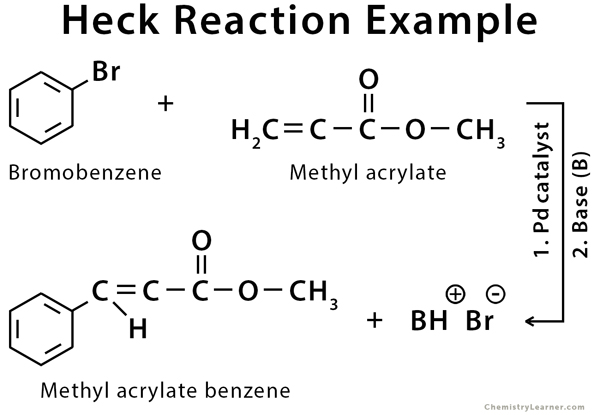
Example of Heck Reaction
The Heck reaction can be used for any alkyl, aryl, or vinyl group. The halide can be chloro (Cl), bromo (Br), or iodo (I) [1,4,5].
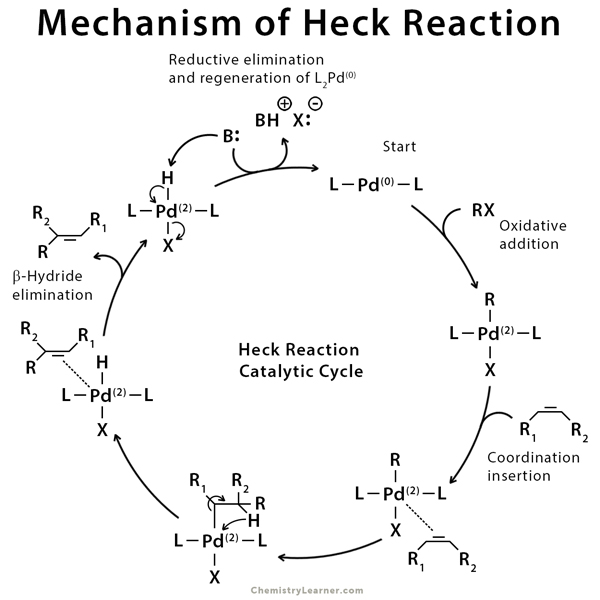
Mechanism of Heck Reaction [3, 6-8]
A commonly accepted mechanism can explain the Heck reaction based on the Pd(0)/Pd(2) catalytic cycle. This mechanistic model gives a better understanding of the scope and limitations of the Heck reaction [3].
Applications of Heck Reaction
The Heck reaction has great significance for both academic and industrial research and in the production of several compounds on a large industrial scale. Fine chemicals, including pharmaceuticals, agricultural chemicals, and high-tech materials, benefit society when appropriately designed. Besides, Heck reaction adds to the new reactivities, increases product selectivity, and reduces volatile organic consumption. This reaction can lead to a substantial breakthrough in industrial research [3].
References
- Definition, examples, and mechanism – Chem.libretexts.org
- Definition and mechanism – Name-reaction.com
- Definition, mechanism, and applications – Mdpi.com
- Examples – Faculty.chemistry.harvard.edu
- Examples and mechanism- Organic-chemistry.org
- Mechanism – Technology.matthey.com
- Mechanism – Chtf.stuba.sk
- Mechanism – Organicreactions.org