Hell-Volhard-Zelinsky Reaction
Explanation: What is Hell-Volhard-Zelinsky Reaction?
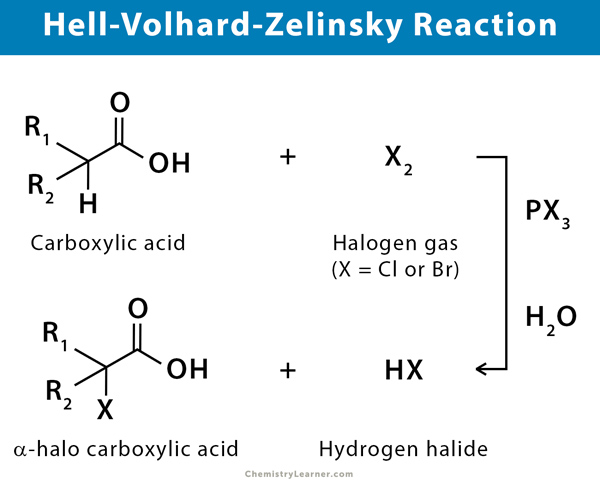
The Hell-Volhard-Zelinsky (HVZ) reaction is an organic reaction that is used to convert a carboxylic acid consisting of alpha hydrogen to an α-halo carboxylic acid, using a phosphorous catalyst, halogen gas, and water. It is a type of substitution reaction. Typically, red phosphorus and bromine are treated with the carboxylic acid followed by hydrolysis to form an α-bromo carboxylic acid. An application of this reaction is in the preparation of α-bromo thioesters [1-4].
The reaction is named after three chemists, the two German chemists Carl Magnus von Hell and Jacob Volhard and the Russian chemist Nikolay Zelinsky. The history of the reaction goes back to 1881 when the first publication by von Hell came out.
Examples of Hell-Volhard-Zelinsky Reaction
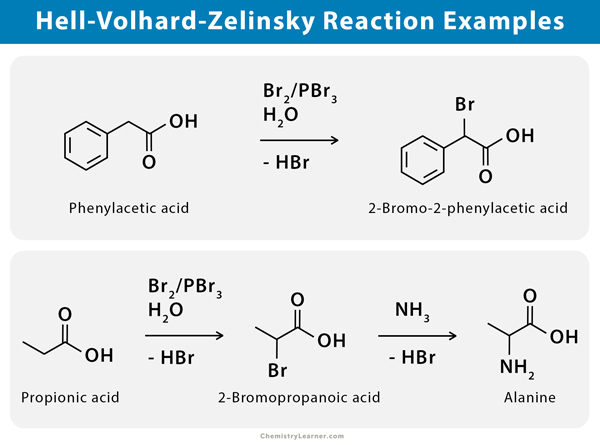
Hell-Volhard-Zelinsky reaction is used to convert phenylacetic acid to 2-bromo-2-phenylacetic acid when reacted with PBr3. HVZ reaction is also used to prepare alanine, an α-amino acid, after the ammonolysis of 2-bromopropanoic acid [1].
Mechanism of Hell-Volhard-Zelinsky Reaction
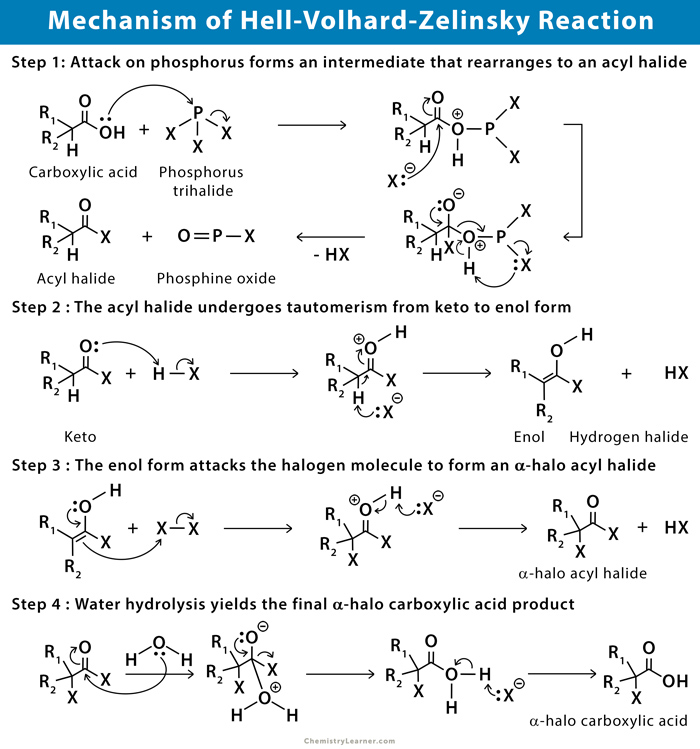
In the first step, the carbonyl oxygen reacts with phosphorus trihalide to form the P-O bond and releases the halide anion. Halide attacks carbonyl group, which creates an intermediate that undergoes rearrangement to release acyl halide, hydrogen halide acid, and phosphine oxide. Further tautomerization of the acyl halide occurs, which forms α-halo acyl halide. Finally, hydrolysis takes place, which leads to the formation of α-halo carboxylic acid [2-4].
References
- Definition and example – Chem.ucla.edu
- Definition and mechanism – Chem.libretexts.org
- Definition and mechanism – Slideshare.net
- Definition and mechanism – Name-reaction.com