Henderson-Hasselbalch Equation
The Henderson-Hasselbalch equation is an equation used in chemistry and biochemistry to determine the pH value of a buffer solution during the dissociation of a weak acid. A weak acid does not dissociate completely. A property of the buffer solution is that the mixture can resist changes in pH when a small amount of acid is added [1-5].
How to Use Henderson Hasselbalch Equation
The Henderson-Hasselbalch equation is used to determine the amount of acid and conjugate base needed to make a buffer solution of a certain pH. Therefore, it is often used to perform the calculations necessary to prepare buffers for use in the laboratory or other applications.
General Formula of the Henderson-Hasselbalch Equation
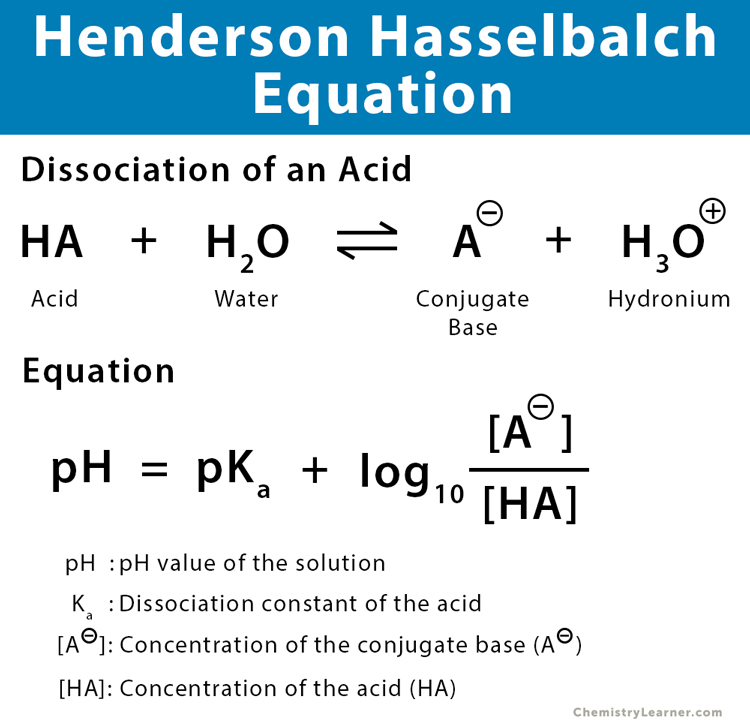
A simple buffer solution consists of a weak acid solution and a salt of the acid’s conjugate base. The equation is given by,
pH = pKa + log10[conjugate base/weak acid]
where,
pH: pH value
Ka: acid dissociation constant
[conjugate base]: Concentration of the conjugate base in moles per liter
[weak acid]: Concentration of the acid in moles per liter
American chemist Lawrence Joseph Henderson first derived the equation in 1908 and, later, by Danish chemist Karl Albert Hasselbalch in 1917.
Derivation of Henderson-Hasselbalch Equation
The Henderson-Hasselbalch equation can be derived from the equilibrium constant expression for the dissociation of a weak acid. Consider the dissociation of the acid HA in water H2O [2-6].
HA + H2O ⇄ A– + H3O+
The acid dissociation constant is given by,
Ka = [A–] [H3O+]/[HA]
or, [H3O+] = Ka[HA]/ [A–]
or, log10 [H3O+] = log10 (Ka[HA]/ [A–])
or, log10 [H3O+] = log10 Ka + log10 ([HA]/[A–])
or, – log10 [H3O+] = – log10 Ka – log10 ([HA]/[A–])
or, pH = pKa + log10 ([A–]/[HA])
From the above equation, the following points can be noted.
1. The value of [A–]/[HA] is dependent on the values of the pH and pKa.
- When pH < pKa, [A–]/[HA] < 1.
- When pH > pKa, [A–]/[HA] > 1.
2. When the pH changes relatively to pKa by one unit, then pH – pKa = 1. Therefore,
1 = log10 ([A–]/[HA])
or, [A–]/[HA] = 10
Thus, the ratio of the dissociated form to the associated form changes by a factor of 10.
3. When the acid is 50% dissociated, the acid and the conjugate base concentrations are the same.
[HA] = [A–]
or, [HA]/[A–] = 1
Therefore,
pH = pKa
The pH value of the solution is equal to the pKa value of the acid.
Henderson-Hasselbalch Equation for Base
The Henderson-Hasselbalch equation also applies to a weak base. A weak base dissociates as follows,
B + H2O ⇄ OH– + HB+
The Henderson-Hasselbalch equation can be derived using the same principles as that for acid and gives,
pOH = pKb + log10 ([HB+]/[B])
where,
pOH: pOH value
Kb: base dissociation constant
[HB+]: Concentration of the conjugate acid in moles per liter
[B]: Concentration of the base in moles per liter
Limitations of Henderson-Hasselbalch Equation
- Fails to predict the pH values of strong acids because it assumes that the acid and its conjugate base concentrations remain constant at equilibrium.
- Can only be applied to a polybasic acid if its consecutive pK values differ by at least 3.
- Fails to give an accurate pH value for extremely buffer solutions.
- Does not consider the self-dissociation by water.
Examples and Problems
P.1. The dissociation of acetic acid (CH3CO2H) in acetate ion (CH3CO2−) is given as follows:
CH3CO2H + H2O ⇄ CH3CO2− + H3O+
Calculate the pH of a buffer solution made from 0.25 M of CH3CO2H and 0.45 M of CH3CO2− that has an acid dissociation constant for CH3CO2H of 1.76 x 10-5 at 25 ͦC.
Soln. Given,
[CH3CO2H] = 0.25 M
[CH3CO2−] = 0.45 M
Ka = 1.76 x 10-5
The Henderson-Hasselbalch equation is given by,
pH = pKa + log10 ([A–]/[HA])
Therefore,
pH = -log10 (1.76 x 10-5) + log10 (0.45 M /0.25 M) = 5
P.2. How much sodium formate (HCOONa), 68.0069 g/mol) do you need to add to 500 mL of 0.50 M formic acid (HCOONa) for a pH 3 buffer. Ka = 1.77 x 10¯4.
Soln. Given,
[HCOONa] = 0.5 ML-1
pH = 3
Ka = 1.77 x 10¯4
The Henderson-Hasselbalch equation is given by,
pH = pKa + log10 ([A–]/[HA])
Therefore,
3 = – log10 (-1.77 x 10¯4) + log10 ([A–]/0.5)
or, 3 = 3.752 + log10 ([A–]/0.5)
or, log10 ([A–]/0.5) = -0.752
or, [A–]/0.5 = 0.177
or, [A–] = 0.177 x 0.5 = 0.0885 ML-1
The relationship between the concentration and the molecular weight is,
Concentration x volume = weight/molecular weight
0.0885 ML-1 x 0.5 L = w/ 68.0069 gM-1
Or, w = 0.0885 ML-1 x 0.5 L x 68.0069 gM-1 = 3 g






Very good formula in chemistry and biochemistry.