Huckel’s Rule
Definition: What is Huckel’s Rule?
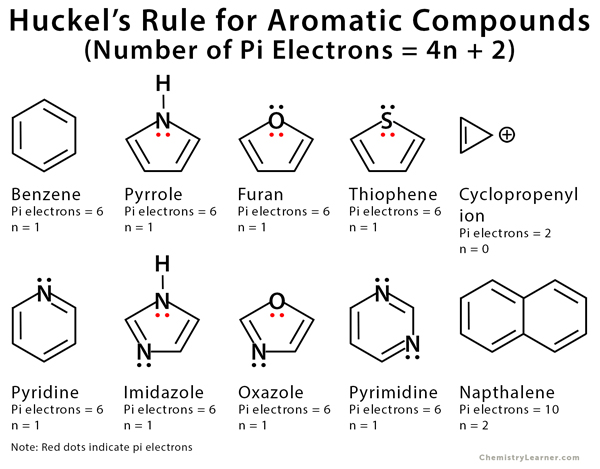
Pi electrons are defined as electrons that participate in pi bonds or lone pairs within p orbitals. In order to be aromatic, a molecule must have a certain number of pi electrons within a closed loop of parallel, adjacent p orbitals. The pi-electron count is defined by the series of numbers generated from 4n+2 where n = zero or any positive integer (i.e., n = 0, 1, 2, etc.). This rule is known as Huckel’s rule. It is used to identify the aromaticity of the ring-shaped planer molecule or ion. The most common case is six pi electrons (n = 1), which is found in benzene, pyrrole, furan, and pyridine [1-7].
The history of the Huckel’s rule goes back to 1931. German physicist and physical chemist Erich Huckel proposed a theory to determine if a planar ring molecule would have aromatic properties.
Criteria for Aromaticity
For a molecule to be aromatic, the following criteria are required [2]:
- The molecule is cyclic (a ring of atoms).
- The molecule is planar (all atoms in the molecule lie in the same plane).
- The molecule is fully conjugated (p orbitals at every atom in the ring).
- The molecule has 4n+2 pi electrons (n=0 or any positive integer).
If a molecule does not satisfy any of the rules 1-3, it is considered nonaromatic. Huckel’s rule does not apply to nonaromatic molecules.
Examples of Aromatic Compounds
Aromatic molecules can be both homocyclic and heterocyclic. Heterocyclic molecules have atoms other than carbon in their molecular structure (eg., furan) [1].
Antiaromatic Compounds
A molecule can be antiaromatic if it follows the first three criteria listed above. However, the fourth criterion is as follows [2]:
- The molecule has 4n pi electrons (n=1 or any positive integer).
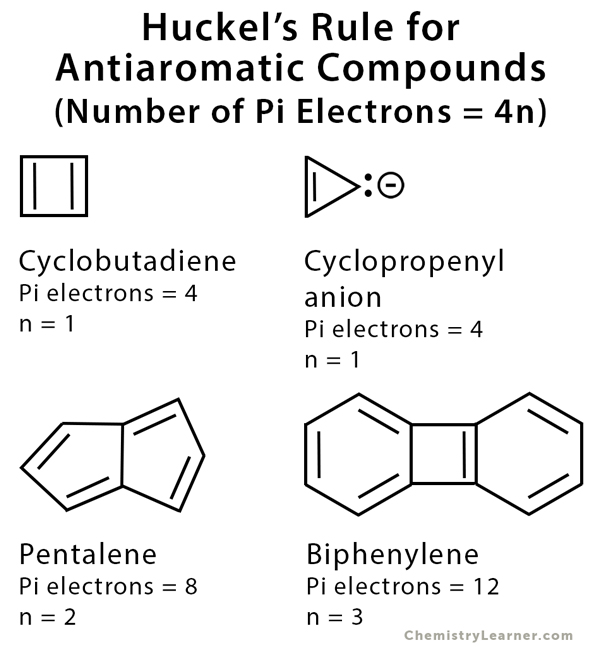
Examples of Antiaromatic Compounds
Cyclobutadiene, pentalene, and cyclooctatetraene are examples of antiaromatic molecules. Cyclopropenyl anion is also antiaromatic because the carbanion can resonate with the double bond, which means the molecule is fully conjugated [2].





good work
Thanku soo much for providing useful data about the topic
ThankYou so much!