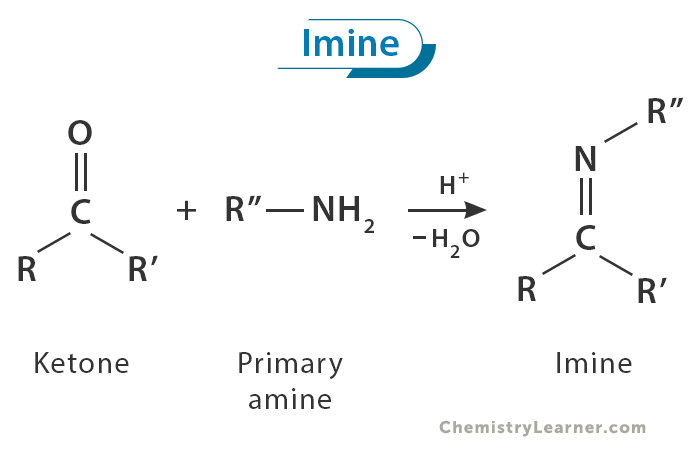
Imine
In organic chemistry, imine is a chemical compound whose structure contains a carbon-nitrogen double bond (C=N), with the nitrogen atom bonded to at least one other carbon atom. The carbon atom is also bonded to two other atoms or groups. A Schiff base is considered a subclass of imines. [1-4]
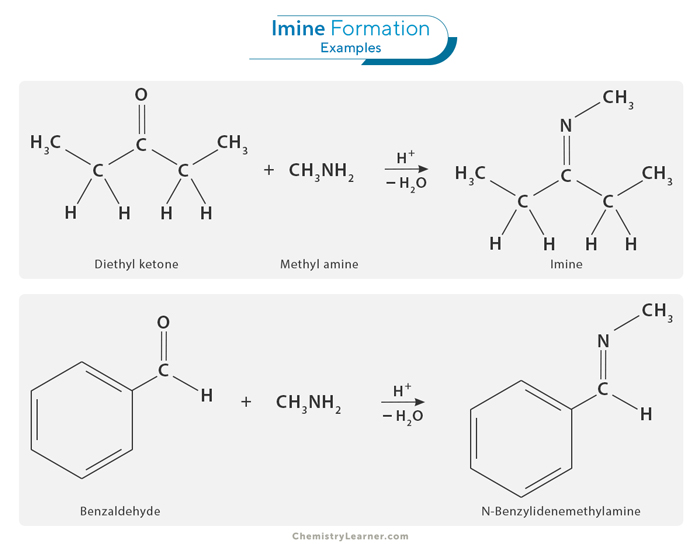
Imine Formation
One of the most common methods for imine synthesis is the condensation reaction between an amine and an aldehyde or ketone. This reaction is typically catalyzed by an acid or base, which helps facilitate the formation of the imine bond. [1-5]
The condensation process occurs with remarkable speed, resulting in the prompt formation of water. Subsequently, the generated water is eliminated through distillation, producing a high-yield, crystalline imine product.
The pivotal step in imine synthesis is eliminating water from the reaction. Without efficient water removal, the imine may revert to its initial aldehyde or ketone form. Thus, imine formation is a reversible process.
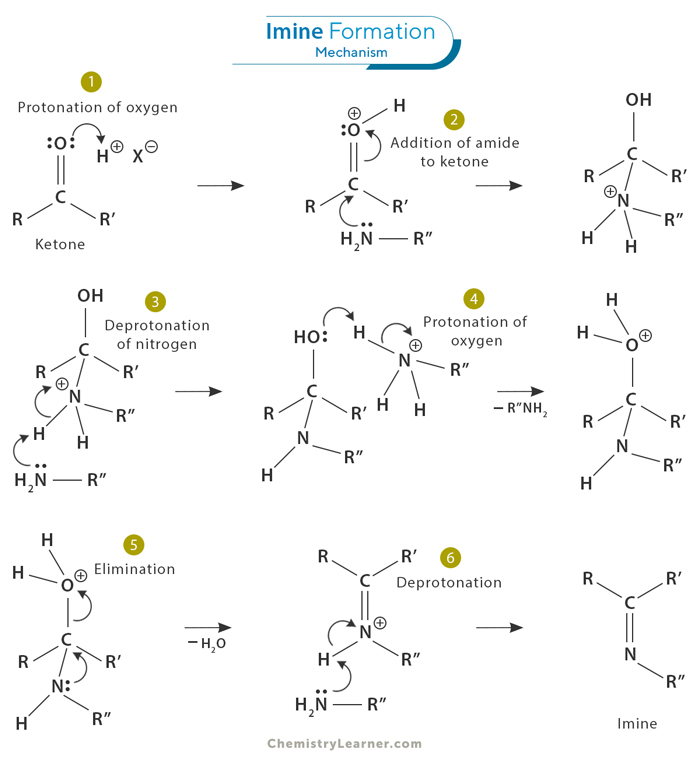
Imine Formation Mechanism
The formation of imines involves a specific mechanism that can be broken down into several steps, shown as steps 1 to 6 in the image. Here is a summary of the mechanism: [1-5]
The amine-aldehyde/ketone reaction initiates with the interaction between a primary amine and an aldehyde or ketone compound. This reaction is characterized by the amine’s nucleophilic nitrogen atom attacking the carbonyl group’s electrophilic carbon atom in the aldehyde or ketone. As a result, a hemiaminal intermediate is formed, which serves as an essential intermediate for subsequent chemical transformations in various organic synthesis pathways.
After the initial step of this chemical reaction, the intermediate undergoes intramolecular dehydration. During this stage, water molecules are effectively removed from the intermediate molecule. This elimination process ultimately leads to the creation of an imine compound. A proton is transferred from the nitrogen atom to an adjacent oxygen atom to achieve this transformation. This proton transfer event triggers water expulsion and facilitates stabilizing the resulting imine product.
Reactions Involving Imines
Imines participate in various reactions that are of great interest to chemists. Two significant reactions include hydrolysis and reduction. [1,4,6]
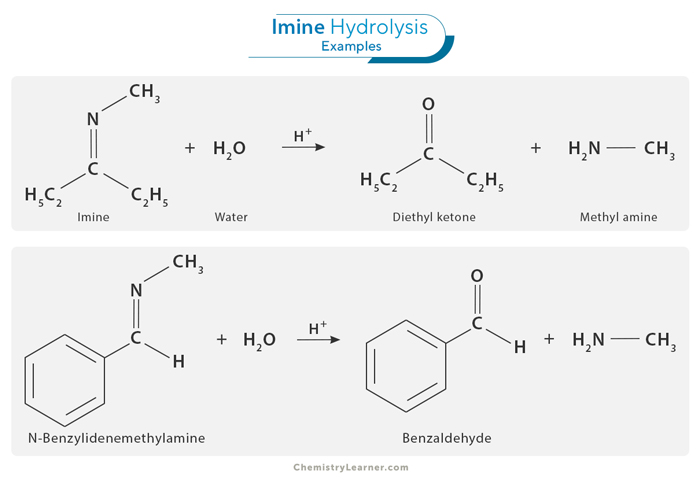
Hydrolysis
One important class of reactions involving imines is hydrolysis. Hydrolysis reactions involve cleavage of the carbon-nitrogen double bond in an imine by adding water, forming an amine and aldehyde or ketone. This process can be achieved under acidic or basic conditions, with each condition leading to different outcomes. In the acidic hydrolysis of imines, a proton (H+) is added to the nitrogen atom, followed by a nucleophilic attack of water on the electrophilic carbon, forming the corresponding amine and carbonyl compound. In basic hydrolysis, hydroxide ions (OH–) initiate the reaction, breaking the C=N bond and producing the amine and a hydroxide salt. The mechanism of imine hydrolysis is of significant importance in organic synthesis. It plays a crucial role in the breakdown of imine-containing compounds in various chemical and biological processes.
Reduction
The reduction of imines involves the addition of hydrogen atoms to the carbon-nitrogen double bond (C=N), resulting in the formation of primary amines. This reaction typically uses reducing agents such as sodium borohydride (NaBH4) or lithium aluminum hydride (LiAlH4). The reduction mechanism begins with the attack of the reducing agent on the imine’s carbon atom, followed by the transfer of hydride ions to the nitrogen atom. This process ultimately converts the C=N bond into a C-N single bond. Imine reduction is a valuable transformation in organic chemistry, as it allows for the synthesis of primary amines, which are essential building blocks for various organic compounds, including pharmaceuticals and complex organic molecules.
Significance
Imine formation and hydrolysis play a crucial role in biological chemistry, with imines being utilized by numerous enzymes as integral components of their reaction mechanisms. Notably, a significant biological reaction involves the establishment of an imine linkage between the carbonyl group of an amino acid and the amine of lysine. [6]
The nitrogen within the C=N bond can easily undergo protonation, forming a protonated imine. This protonated imine functions as an electron-accepting moiety. When part of an adduct, it can draw electrons away from the bonds within the connected molecule. It results in bond dissociation, a crucial process in various metabolic reactions, such as glycolysis and amino acid metabolism.





you need to add the meanings of each word for ine, ane, one, nye, ect. at the end of a word
These are IUPAC nomenclatures