Isobaric Process
An isobaric process is a thermodynamic process that takes place at constant pressure. The term isobaric originates from two Greek words – iso, meaning equal, and baros meaning pressure [1-4].
Examples
Suppose an ideal gas is confined to a container. Constant pressure is achieved by allowing its volume to expand or contract when heat is transferred between the gas and the surroundings. Any change in pressure is adjusted by changing the volume. This process of expansion and contraction is illustrated in the given diagram of a weighted piston [1-4].
Some other examples of isobaric processes in real life are:
- During boiling water to steam or freezing water to ice, water either expands or contracts to maintain constant pressure. The heat supplied during boiling increases the temperature and does external work.
- The operations of heat engines often involve isobaric processes. Heat engines operate on thermodynamic cycles that take heat from a hot reservoir, reject heat to a cold reservoir, and perform valuable work. Specific steps in the Brayton and Rankine cycles take place under isobaric conditions.
Heat Transfer in an Isobaric Process
Isobaric processes can be further understood using the first law of thermodynamics. It gives a relationship between the heat transfer (Q) between the system and the surroundings, the internal energy change (ΔU) in the system, and the work done (W). The mathematical representation of the first law is [1-4]
ΔU = Q – W
The work done is given by the product of pressure (p) and change in volume (ΔV). It is known as boundary work.
W = pΔV
A detailed explanation of the work done is discussed in the next section.
A part of the transferred heat will change the internal energy. Therefore, we use the system’s enthalpy change to determine the heat. The enthalpy (H) is given by
H = U + pV
Or, ΔH = ΔU + pΔV +VΔp
Putting this in the classical expression of the first law, we get
ΔH = Q – pΔV + pΔV +VΔp
or, ΔH = Q + VΔp
For an isobaric process, Δp = 0.
ΔH = Q
or, Q = (H2 – H1)
Therefore, the heat transferred is the change in the system’s enthalpy. The above equation is the isobaric form of the first law. It is calculated for open systems.
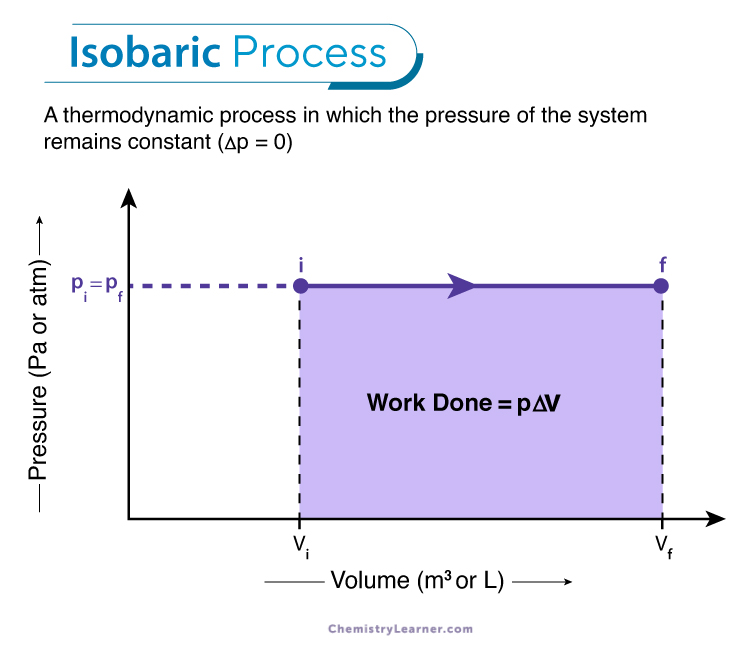
Work Done in an Isobaric Process
The work done in an isobaric process can be calculated from the P-V diagram. A P-V diagram is a graphical representation of the relationship between the pressure and the volume. Since the pressure remains constant in an isobaric process, the P-V diagram is a straight line parallel to the volume axis, as shown below. [1-4]
The work done is given by integrating the area under the p-V line.
W = ∫pdV
or, W = p ΔV
or, W = p (Vf -Vi)
Where
Vi: initial volume of the gas
Vf: final volume of the gas
The expansion or contraction of the gas in an isobaric process is accompanied by a change in temperature. It is often convenient to calculate the work in terms of the temperature since the latter can be measured easily. We will use the ideal gas equation below for this purpose.
PV = nRT
Where
n: number of moles of the gas
R: universal gas constant
At constant pressure, we can write the above equation as
pΔV = nRΔT
Or, p(Vf – Vi) = nR(Tf – Ti)
Or, W = nR(Tf – Ti)
Where,
Ti: initial temperature of the gas
Tf: final temperature of the gas
Sign Convention
Recall the expression for work done when the gas expands or contracts.
W = p ΔV
The sign of work done depends on whether ΔV can be positive or negative. Consequently, there can be two outcomes. [1]
- When the gas expands, its final volume is greater than the initial volume, i.e., Vf >Vi. Therefore, ΔV > 0 and W > 0. It means that the work done by the gas is positive.
- When the gas contracts, its final volume is less than the initial volume, i.e., Vf < Vi. Therefore, ΔV < 0 and W < 0. It means that the work done by the gas is negative.
Example Problems
Problem 1: 2 m3 volume of an ideal gas is stored under a constant pressure of 4 × 105 N/m2. The temperature is increased till it expands to triple its initial volume. What is the work done?
Solution:
Given: Vi = 2m3 p = 4 × 105 N/m2 Vf = 3Vi
The work done is
W = p(Vf – Vi) = p(3Vi – Vi)
= (4 × 105 N/m2)(6m3 – 2m3)
= (4 × 105 N/m2) × 4m3
= 16 × 105 J or 1.6 × 106 J
Problem 2: A gas is kept in a container at 0.5 atm of constant pressure. It is compressed to half its initial volume of 200L. Calculate the work done and justify the sign convention.
Solution:
Given: p = 0.5 atm Vi = 100L Vf = ½ Vi
The work done is
W = p(Vf – Vi) = p( ½ Vi – Vi)
= 0.5 atm × (100L – 200L)
= (0.5 × 101325 N/m2)(-100 × 10-3 m3)
= 50662.5 N/m2 × (-) 0.1 m3
= -5066.25 J or -5.06625 kJ
Here, compression occurs, so ΔV is negative. Therefore, the net work done will also be negative.






In case of steam drum in a boiler specific volume changes and volume does not change, why is it said heat is added at constant pressure, and work flow is also mentioned and W = 0, pv work is important. Could u explain why is it a constant pressure process when Delta V = 0?
Thank u
The phase change from water to steam occurs at the pressure of the steam drum. No macroscopic work is done by the steam drum as a whole system, but there are microscopic pressure-volume work interactions at the phase boundary. The system is described as a constant pressure process due to the regulation of pressure and the ongoing phase change within it.