Le Chatelier’s Principle
Table of Contents
Le Chatelier’s Principle states that if a dynamic equilibrium is disturbed by changing the conditions, the equilibrium shifts in the direction that counteracts the change. It applies to a chemical reaction at equilibrium that is disturbed by altering the concentration, pressure/volume, or temperature. An application of Le Chatelier’s Principle is Haber’s process for synthesizing ammonia [1-4].
The law is named after French chemist Henry-Louis Le Chatelier, who presented his results on chemical equilibrium in 1884.
1. Change of Concentration
Increasing the reactants’ concentration will shift the equilibrium to produce more products. Increasing the products’ concentration will shift the reaction to make more reactants. Decreasing reactants favors reactants. Decreasing product favors products [1-6].
Consider a chemical equilibrium between four substances, A, B, C, and D, as shown below.
A + 2B ⇋ C + D
Case 1
Suppose we increase the concentration of A. The equilibrium’s position will move in such a direction that the system will reduce the amount of A by reacting it with B to produce more C and D. Thus, the reaction will shift to the right and reestablish a new equilibrium.
Case 2
Now, suppose we decrease the concentration of A. The equilibrium’s position will move in the direction of increasing A. It means that C and D will react to reproduce A. Thus, the equilibrium’s position moves towards the left.
Case 3
Suppose we reduce the concentration of one of the products, say C. The equilibrium’s position will move right to produce more C. If we continue to remove C, the equilibrium will shift further right until we get an irreversible reaction.
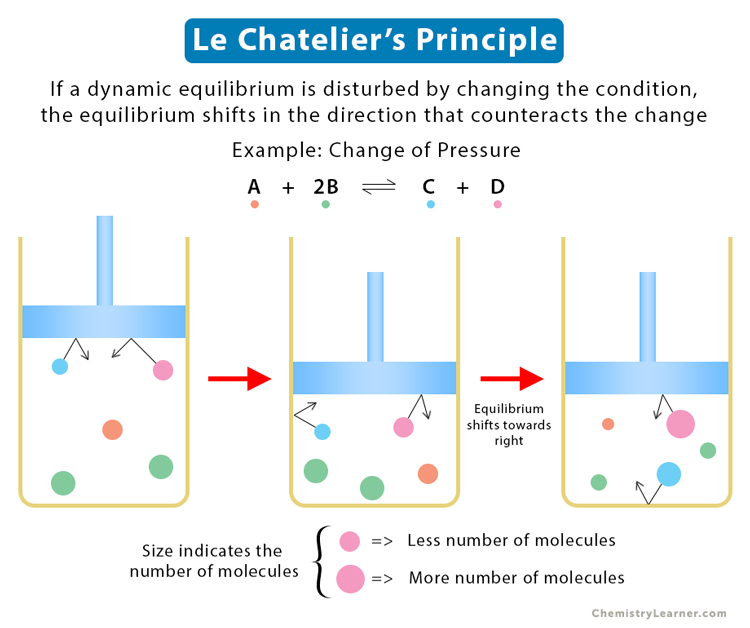
2. Change of Pressure
Le Chatelier’s Principle for change of pressure applies when the substances are in their gaseous state. Consider the following reaction involving three molecules of the reactants A and B and two molecules of the products C and D [1-6].
A (g) + 2B (g) ⇋ C (g) + D (g)
Case 1
Suppose the pressure is increased. The equilibrium’s position will move so that the pressure is reduced to what it was before. Pressure is caused by the gas molecules hitting the walls of the container—the higher the number of molecules, the greater the pressure. So, by decreasing the number of molecules, the pressure can be reduced. In this case, the system will respond to produce fewer molecules.
In the above reaction, there are three moles on the left and two on the right. The system will reduce the number of molecules on the left by creating more C and D. Therefore, increasing the pressure shifts the equilibrium to the side of fewer moles of the gas molecules.
Case 2
Suppose the pressure is decreased. The equilibrium will move in the direction of increasing pressure. It can happen only when the reaction produces more moles of the gas, which is happening on the left. Therefore, the equilibrium will shift left.
Case 3
Suppose there is an equal number of molecules on both sides. Then, altering the pressure will not affect the reaction since the equilibrium will shift neither left nor right.
The effect of volume is opposite to that of pressure.
3. Change of Temperature
To understand the effect of temperature, we must know if the forward chemical reaction is exothermic (heat released) or endothermic (heat absorbed). If the forward reaction is exothermic, the reverse reaction is endothermic, and vice versa [1-6].
Let us consider the reaction between A and B to produce C and D. Suppose the reaction liberates heat (exothermic) in the forward direction. The enthalpy of reaction (ΔH) will be negative.
A (g) + 2B (g) ⇋ C (g) + D (g) ΔH is negative
Heat may be added to a system externally or produced due to the reaction. For an exothermic reaction, heat is considered a product of the reaction. For an endothermic, heat is considered a reactant. So, increasing or decreasing temperature can be considered the same as increasing or decreasing the reactants’ or products’ concentration.
Case 1
Suppose the temperature is increased. Then, the system will try to reduce the temperature. It is possible when the heat is absorbed. We know that the reverse reaction is endothermic. Therefore, the equilibrium shifts toward the left. The new equilibrium position is such that the reaction contains more A and B and less C and D.
Case 2
Suppose the temperature is decreased. Then, the equilibrium shifts in a direction to produce more heat. It is only possible by facilitating the forward reaction, which is exothermic. Therefore, decreasing the temperature favors the production of C and D.
To summarize
- Increasing the temperature of a system in dynamic equilibrium favors the endothermic reaction.
- Decreasing the temperature of a system in dynamic equilibrium favors the exothermic reaction.
Le Chatelier’s Principle Example
As shown below, let us consider the complex ion formation between cobalt (Co2+) ions and water (H2O) molecules or chloride ions.
[Co(H2O)6]2+ (aq., pink) + 4 Cl– (aq.) + heat ↔ [CoCl4]2– (aq., blue) + 6 H2O (l)
The color of the complex ion formed between Co2+ and H2O is pink. When hydrochloric acid (HCl) is added, the solution turns blue. It is due to forming a charged coordination complex between Co2+ and chloride ions (Cl–) present in aqueous HCl. This reaction is reversible and rapidly reaches a point of chemical equilibrium, which is evident by the color of the solution.
Le Chatelier’s Principle and Adding a Catalyst
A catalyst is a substance that increases the reaction’s speed. The addition of a catalyst has no impact on the equilibrium’s position. The reason is that catalyst speeds up the rates of both the forward and reverse reactions. The rates remain equal, as both change by the same amount [1,2,4,6].