Malonic Ester Synthesis
Definition: What is Malonic Ester Synthesis?
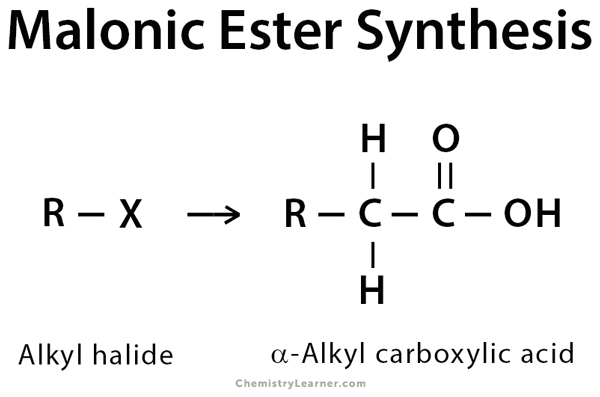
Malonic ester is a diester molecule that can undergo hydrolysis to produce 1,3-dicarboxylic acid. The dicarboxylic acid, if gently heated, can undergo a decarboxylation reaction to produce carbon dioxide and an enol molecule. The enol then interconverts into a more stable carboxylic acid, like acetic acid. This reaction is commonly known as malonic ester synthesis. This method conveniently allows the synthesis of mono- and disubstituted acetic acids, i.e., carboxylic acids bearing one or two alkyl groups on the α-carbon. The α-carbon on the malonic ester is relatively more acidic as compared to other esters. Alkylation at the alpha position can, therefore, be carried out with an alkyl halide (R-X) and relatively mild bases such as sodium hydroxide (NaOH) or sodium ethoxide (NaOEt) [1-7].
Examples of Malonic Ester Synthesis
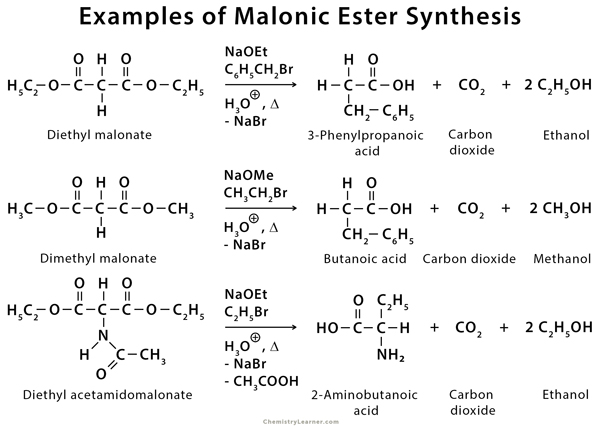
The most commonly used malonic ester is diethyl malonate, also known as diethyl propanedioate. This method can also be used to synthesize amino acids [1-6,8].
Mechanism of Malonic Ester Synthesis
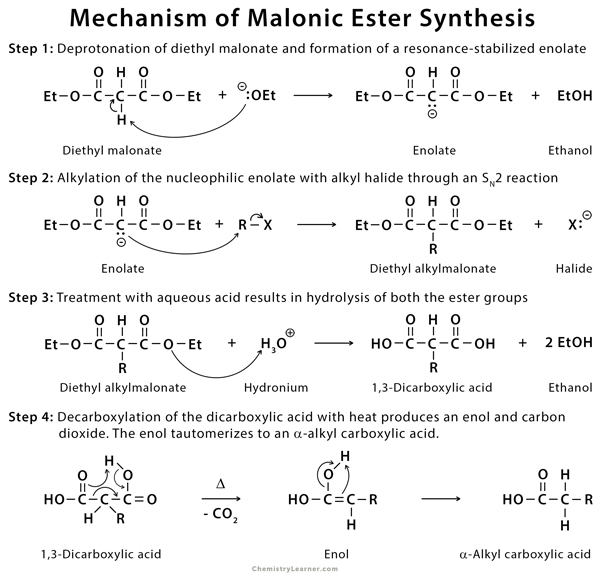
Malonic ester is an active methylene compound. The sequence of reactions is as follows [1-3,7]:
- Deprotonation of the ester to form an enolate
- Alkylation of an active methylene enolate
- Hydrolysis of the esters to a dicarboxylic acid
- Decarboxylation of a 1,3-dicarboxylic acid to give an enol which tautomerizes to α-alkyl carboxylic acid
Applications of Malonic Ester Synthesis
Malonic ester has been found useful in organic synthesis. For example, condensing urea with diethyl malonate produces barbituric acid. A large variety of barbiturate drugs is used for the treatment of insomnia and anxiety [9].
References
- Definition, example, and mechanism – Chem.libretexts.org
- Definition, example, and mechanism – Chem.ucalgary.ca
- Definition, example, and mechanism – Uobabylon.edu.iq
- Definition and example – Chem.ucla.edu
- Definition and example – Ochempal.org
- Definition and example – Askthenerd.com
- Definition and mechanism – Pharmaxchange.info
- Examples – Organic-chemistry.org
- Applications – Onlinelibrary.wiley.com