Marquis Reagent
Definition: What is Marquis Reagent?
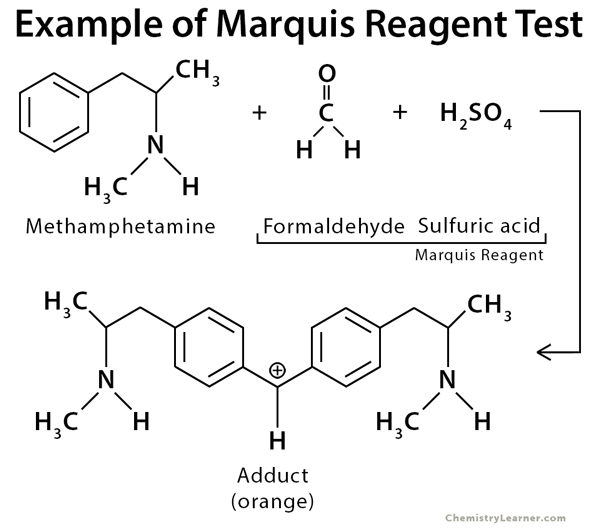
Marquis reagent is composed of a mixture of formaldehyde and concentrated sulfuric acid. It is used as a simple spot-test to identify illicit drugs belonging to the alkaloid family and other compounds. Alkaloids are natural substances typically containing basic nitrogen atoms. The test employed for this purpose is called the Marquis reagent test. Many recreational drugs like ecstasy, heroin, and amphetamines can be detected in this test [1-4].
The history of this reagent goes back to 1896 when Russian chemist Eduard Marquis discovered it.
Preparation of Marquis Reagent
Marquis reagent can be prepared by adding 100 mL of concentrated (95–98%) sulfuric acid to 5 mL of 40% (v/v) formaldehyde. The reagent should be stored in the freezer for maximum shelf life [1-4].
Principle of Marquis Reagent Test
The test is performed by scraping off a small amount of the alkaloid substance and adding a drop of the bright and colorless reagent. The signature of the alkaloid is both the initial color produced and the sequence of color changes occurring with time. The results are analyzed by observing the color of the resulting mixture and by the time taken for the change in color to become noticeable [1-4].
Results of Marquis Reagent Test
The table below shows the final color produced after Marquis reagent is added to a few common substances. The final color is defined as the color, formed within 1-2 min, that remained after any intermediate colors have disappeared [1-4].
| Substance | Final Color |
| Amphetamine | Intense reddish-orange to dark reddish-brown |
| Aspirin | Deep red |
| Codeine | Very deep purple |
| Heroine | Purple |
| LSD | Olive black |
| Methamphetamine | Deep reddish-orange to dark reddish-brown |
| Morphine | Intense reddish-purple |
| Opium | Dark greyish reddish brown |
| Sugar | Dark brown |





Excellent information I was searching for. Got a very clear concept from the informations provided