McLafferty Rearrangement
Definition: What is McLafferty Rearrangement?
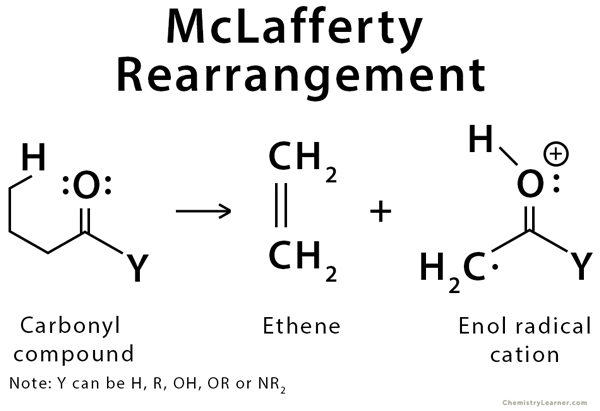
Mass spectrometry is a technique used to determine the mass of a given molecule. The molecule is placed inside the spectrometer and ionized by high-energy electrons. After the molecules are ionized, they become radical cations that can then be sorted and weighed. When hydrogen is available at the gamma position on carbonyl compounds, these compounds undergo a specific rearrangement due to the cleavage of multiple bonds, including the bonds at alpha and gamma positions. This reaction is known as the McLafferty rearrangement. This rearrangement is applied in structural elucidation using mass spectroscopy as the tool.
The rearrangement involves a six-membered ring transition state in which the carbonyl group pulls off the gamma proton, splitting the molecule into two pieces. These pieces consist of an enol radical cation and a neutral alkene fragment. The enol radical cation is observed in the mass spectrum, while the neutral alkene fragment is not observed. Any carbonyl compound with hydrogen in the gamma position will likely have a peak in the mass spectrum corresponding to the enol radical cation formed by the McLafferty rearrangement. Both ketones and aldehydes give prominent molecular ion peaks though the [M+] peak is more prominent in ketones. Only ketones and aldehydes that do not undergo this rearrangement are those which lack a side chain that can donate a hydrogen atom to the pi system [1-5].
This reaction has been named after American chemist Fred McLafferty who published his findings in 1959.
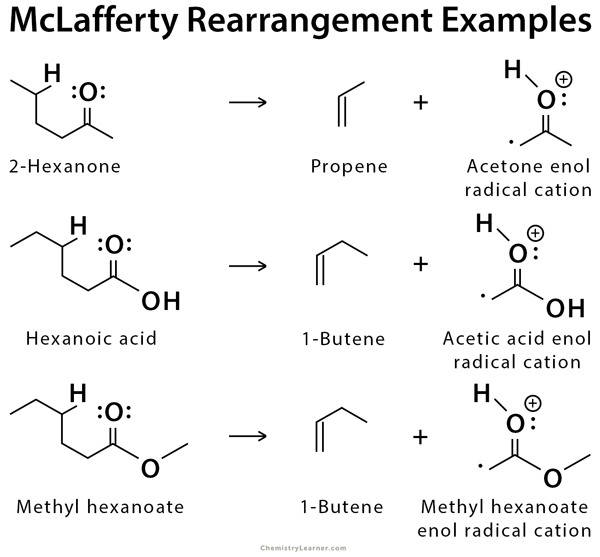
Examples of McLafferty Rearrangement
An example of McLafferty rearrangement is the fragmentation of 2-hexanone, producing acetone enol radical cation and a neutral molecule of propene. Other examples include carboxylic acid and ester [1-4].
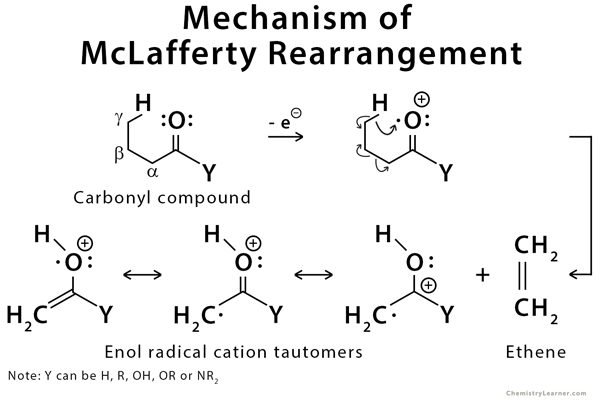
Mechanism of McLafferty Rearrangement
This rearrangement is mediated by the pi systems of the carbonyl group [1-2].
References
- Definition, example, and mechanism – Chem.libretexts.org
- Definition, example, and mechanism – Chem.ucla.edu
- Definition and example – Whitman.edu
- Definition and example – People.whitman.edu
- Definition – Aklectures.com





The information was very helpful. I appreciate it deeply.
Thanks, this was very helpful. BTW Please change the “mass spectroscopy” to “mass spectrometry”.