Newman Projection
Conformations are different structural arrangements of a molecule due to rotation. Visualizing the different conformations of simple hydrocarbons helps us understand their stability and reactivity. Newman projection represents a molecule’s conformational arrangement from a specific viewpoint. Our viewpoint is directed along a particular bond of interest: the carbon-carbon sigma bond in hydrocarbons. The functional groups attached to the carbon atoms reveal their interactions and steric hindrances [1-4].
Newman projection comes from the American chemist Melvin S. Newman, who introduced this concept in the early 20th century.
How to Draw Newman Projections
Drawing Newman projection allows chemists to visualize and analyze the three-dimensional arrangement of atoms in a molecule. Here is a step-by-step guide on how to draw Newman projection: [2,3]
Step 1: Identify the carbon-carbon (C-C) bond you want to visualize in the Newman projection. This bond will be your central axis.
Step 2: Mentally or physically sketch the molecule, ensuring you have a clear picture of the three-dimensional arrangement of atoms around the chosen C-C bond. Imagine looking down the C-C bond from one carbon atom toward the other.
Step 3: Draw a dot enclosed by a circle. The front carbon is closer to your viewpoint and is represented by the dot. The back carbon is further from your viewpoint and is represented by the circle.
Step 4: Position the functional groups on both carbon atoms. Write their chemical symbols on the left, right, top, and bottom of the dot and the circle.
Step 5: Connect the groups to their respective carbon by drawing lines. One set of lines should radially come out of the dot. Another set should be perpendicular to the tangents of the circle.
Examples
To better understand how Newman projection works and its significance in studying molecular conformations, let us explore some practical examples, focusing on two hydrocarbons: ethane and butane [1,2,4-6].
1. Ethane
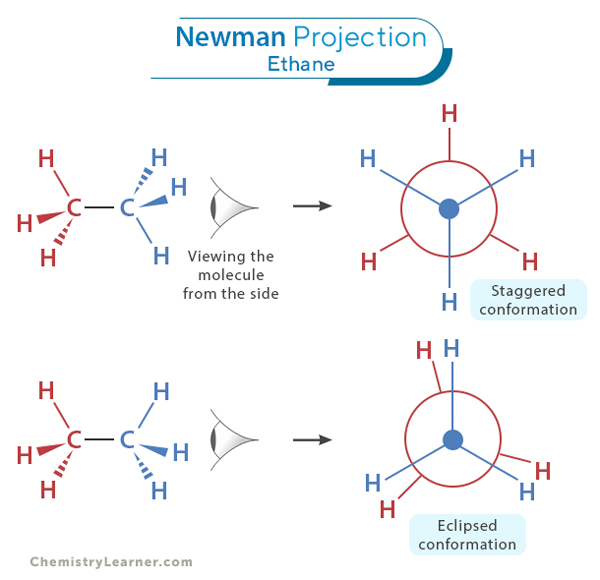
Ethane (C2H6) is the simplest alkane, consisting of two carbon atoms bonded by a single sigma bond and surrounded by hydrogen atoms. It is an excellent starting point to grasp the concept of Newman projection. Ethane exists in two primary conformations: the staggered conformation and the eclipsed conformation.
Staggered Conformation
The hydrogen atoms attached to the two carbon atoms are positioned to maximize the distance between them, as shown in the image. This arrangement minimizes steric hindrance, the repulsion or crowding between the electron cloud surrounding the C-H bonds. As a result, the staggered conformation is the most stable and lowest-energy configuration for ethane. When visualized through a Newman projection, the front carbon, with three hydrogens, and the back carbon, also with three hydrogens, appear staggered or offset.
The angle that describes the rotation of one plane or set of atoms about a common bond axis relative to another plane or set of atoms is called the dihedral angle. The dihedral angle in the staggered configuration is 60 degrees.
Eclipsed Conformation
We get the eclipsed conformation when the staggered conformation is rotated by 60 degrees. Here, the hydrogen atoms are aligned directly on each other. In other words, the C-H bond of the front carbon is in direct alignment with that of the back carbon, resulting in a high-energy, less stable conformation. The Newman projection for this conformation shows the front carbon eclipsing the back carbon with a dihedral angle of 0 degrees.
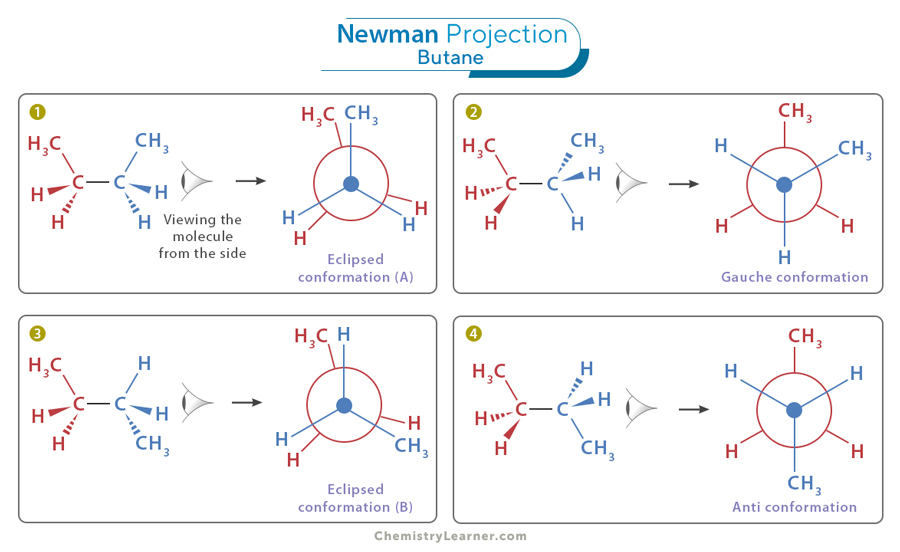
2. Butane
Butane (C4H10) is a slightly more complex hydrocarbon consisting of four carbon and ten hydrogen atoms. It provides an excellent example of how Newman projection can be applied to study the conformations of larger molecules. We will focus our attention on the C2-C3 bond. The various conformations (1, 2, 3, and 4) are shown in the image below.
Staggered Conformation
The staggered conformation of butane is the lower-energy and more stable conformation due to minimized steric hindrance between the two methyl groups. Butane exhibits two types of staggered conformations: the anti (4) and the gauche (2) conformations.
Anti Conformation
The front and back methyl groups are perfectly staggered, with the dihedral angle between them being 180 degrees. This conformation is the most stable due to minimal steric hindrance between the methyl groups.
Gauche Conformation
The front and back methyl groups are staggered in the gauche conformation but not as perfectly as in the anti conformation. The dihedral angle is 60 degrees, resulting in increased steric hindrance between the methyl groups.
Eclipsed Conformation
In eclipsed conformation A (1), the methyl groups appear to be directly on top of each other. In eclipsed conformation B (3), the methyl group of the front carbon is on top of the hydrogen atom of the back carbon.
When the electron clouds encircling the hydrogen atoms in the methyl groups are brought into proximity, electron-electron repulsion occurs, which results in destabilization. Therefore, the eclipsed conformation is higher in energy and less stable because of the more significant steric hindrance between atoms.
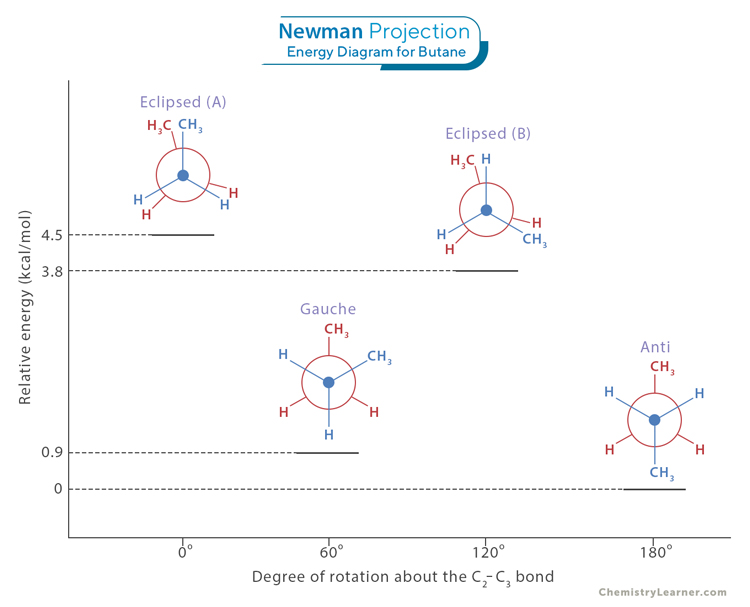
Newman Projection Energy Diagram
Newman projection energy diagrams provide a powerful visual representation of the relative stabilities of different conformations and help elucidate the concept of torsional strain. Examining the energy curve allows us to identify the most stable and least stable conformations. The lowest points on the curve correspond to the most stable conformations. In contrast, the highest points represent the least stable conformations. The image below shows the energy levels of all four conformations of butane [4].
Peaks in the energy curve correspond to conformations with high torsional strain. A torsional strain occurs when groups of adjacent atoms come close to each other, leading to repulsive interactions. These high-energy eclipsed conformations are less stable and less favored in chemical reactions. The staggered conformations typically have lower energy than eclipsed conformations. It reflects the reduced steric hindrance and torsional strain in staggered conformations. Understanding the energy difference between the staggered and the eclipsed conformations is essential in explaining the torsional strain experienced by molecules and the preference for staggered conformations.