Nitrile
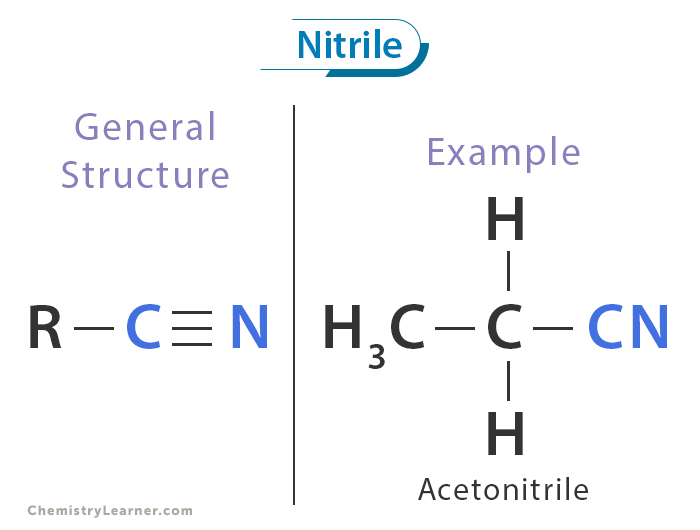
A nitrile is an organic compound that contains a cyano group (-C≡N) attached to an alkyl or aryl group. The cyano group consists of a carbon atom triple-bonded to a nitrogen atom, imparting distinct chemical properties to these compounds. For example, acetonitrile (CH₃CN) is a simple nitrile used as a solvent in organic chemistry.
Nitriles are important in organic chemistry due to their versatility; they can be used as intermediates in the synthesis of various compounds, including amines, carboxylic acids, and amides.[1-4]
Structure
The structure of a nitrile is characterized by a carbon atom triple-bonded to a nitrogen atom (-C≡N). This triple bond gives the nitrile group a linear geometry, with a bond angle of 180° around the carbon-nitrogen triple bond. [1-5]
Electronegativity is a measure of how strongly an atom attracts electrons in a bond. Since nitrogen is more electronegative than carbon, it pulls electrons closer, giving the carbon a partial positive charge and the nitrogen a partial negative charge. This affects how nitriles react with other chemicals.
The linear geometry of the cyano group (-C≡N) contributes to the molecule’s stability and reactivity, making nitriles versatile intermediates in organic synthesis. The strong polarization of the triple bond allows nitriles to engage in various interactions, including hydrogen bonding and coordination with metal ions, which are essential in many chemical reactions.
Synthesis
The synthesis of nitriles involves several key methods, each offering a different way to create these important organic compounds. [1-5]
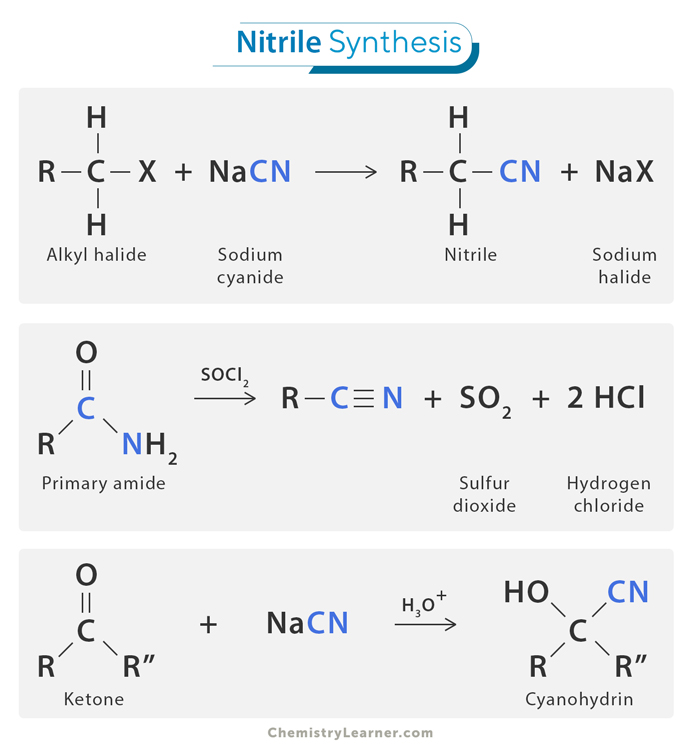
1. Alkylation of Cyanide Salt
One common way to produce nitriles is through the alkylation of a cyanide salt, a type of nucleophilic substitution where the cyanide ion replaces the halide ion in an alkyl halide. This method involves reacting a cyanide salt, like potassium cyanide (KCN) or sodium cyanide (NaCN), with an alkyl halide (a compound where a carbon chain is attached to a halogen atom). During the reaction, the alkyl group from the halide replaces the cyanide ion, forming an alkyl nitrile. This method is often used because it is straightforward and effective.
2. Cyanohydrin Formation
Another important method for synthesizing nitriles is through cyanohydrin formation. This process starts with a carbonyl compound, such as an aldehyde or ketone. When hydrogen cyanide (HCN) or a cyanide ion is added to the carbonyl compound, it forms an intermediate called a cyanohydrin. Afterward, this intermediate undergoes dehydration (removal of water) to create the nitrile group. For example, when acetone (CH₃COCH₃) reacts with hydrogen cyanide (HCN), it forms acetone cyanohydrin (CH₃C(OH)(CN)CH₃), which can be dehydrated to form a nitrile. This method is valuable because it allows for the formation of nitriles from readily available starting materials.
3. Amide Dehydration
A third method to synthesize nitriles is by dehydrating primary amides, which are organic compounds containing the -CONH₂ group. In this process, the primary amide is heated in the presence of a dehydrating agent or catalyst. As the reaction proceeds, water is eliminated, leaving behind the nitrile. This method is particularly useful when starting from amides, which are common organic compounds.
Reactions[1-5]
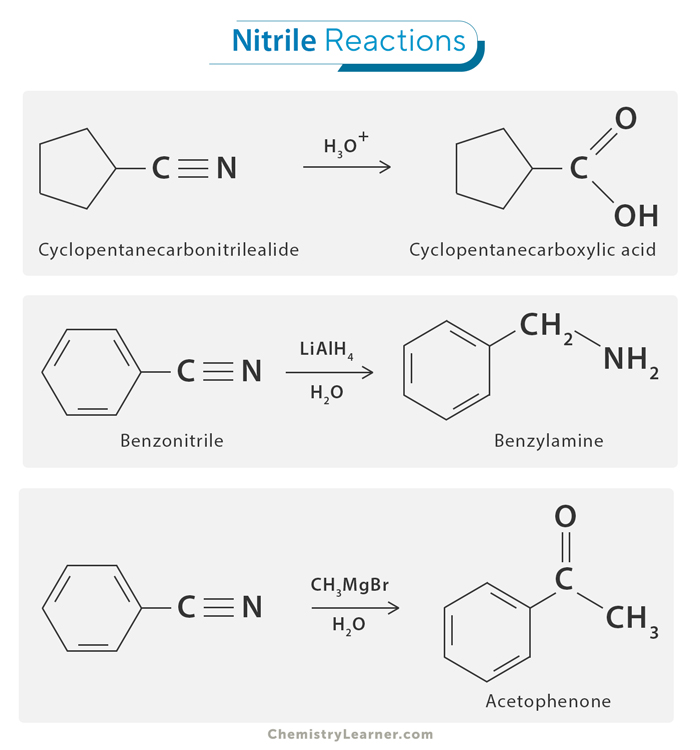
1. Hydrolysis
Hydrolysis of nitriles involves breaking the carbon-nitrogen triple bond (-C≡N) using water to form carboxylic acids or their salts. This reaction usually requires a strong acid or base to help the process. In acidic conditions, the nitrile first gains a hydrogen ion (H⁺), making it easier for a water molecule to attach to the carbon atom. This creates an unstable intermediate that quickly transforms into a carboxylic acid. In basic conditions, hydroxide ions attack the nitrile directly, leading to the formation of a carboxylate ion.
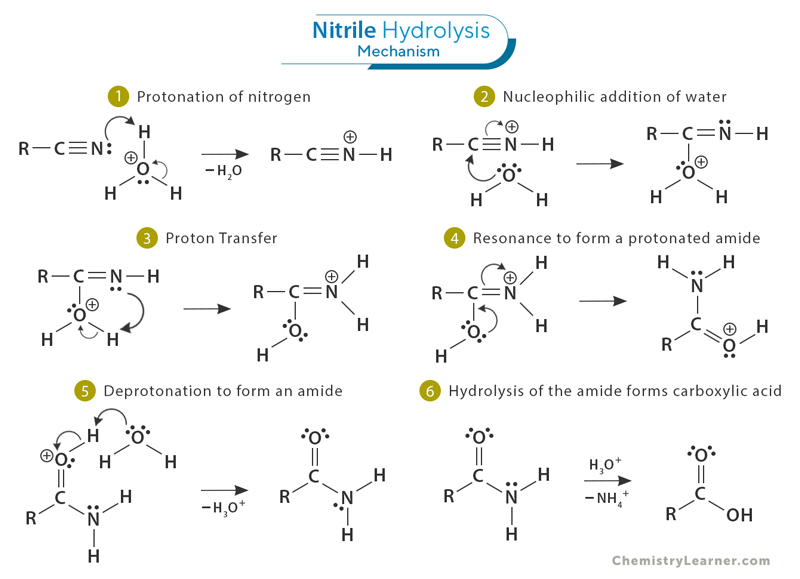
Mechanism
The process starts with the nitrile gaining a proton or hydrogen ion (H⁺) (1), which makes it easier for the less reactive water molecule to attach to the carbon in the nitrile (2). This leads to the formation of a protonated amide, an intermediate (4). Water then removes a proton, creating an amide and regenerating the acid catalyst (5). Finally, the amide undergoes further hydrolysis to produce a carboxylic acid, with the nitrogen being released as ammonium (NH₄⁺) (6).
2. Reduction
Nitriles can be reduced to primary amines using reducing agents like lithium aluminum hydride (LiAlH₄) or hydrogen gas with a catalyst. This process is similar to hydrogenating alkenes, where the carbon-nitrogen triple bond is broken and replaced by single bonds, forming an amine.
3. Reaction with Organometallic Reagents
Organometallic reagents, such as Grignard reagents, are compounds where an alkyl or aryl group is bonded to a magnesium halide. These reagents are highly reactive and can add to the carbon atom in a nitrile group, forming a new carbon-carbon bond. This reaction is known as Grignard reaction. The carbon in the nitrile is electron-deficient and attracts the electron-rich organometallic reagent. This addition forms an imine intermediate, which can then be hydrolyzed or further reacted to produce ketones. This reaction is important in organic synthesis because it allows chemists to build more complex molecules from simpler ones.
Applications
- Nitrile gloves are made from nitrile rubber, which is derived from acrylonitrile. These gloves are essential in medical, laboratory, and industrial environments due to their resistance to chemicals, punctures, and allergens compared to latex gloves.
- Acrylic fibers, produced from polyacrylonitrile, are used in clothing, blankets, and carpets. They are known for being lightweight, warm, and resistant to moths and chemicals.
- Many nitrile compounds serve as intermediates in the synthesis of pharmaceutical drugs. For instance, nitriles are involved in the production of medications for conditions like high blood pressure, depression, and cancer.