PCC Reagent
Definition: What is PCC Reagent?
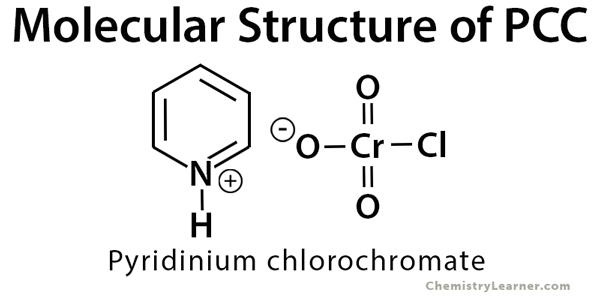
PCC (pyridinium chlorochromate) reagent is used in the organic synthesis of aldehyde and ketone from primary and secondary alcohols, respectively. It is a yellow-orange colored salt with the formula [C5H5NH]+[CrO3Cl]–. PCC oxidizes the alcohol (OH) group and does not affect any other functional group or double bond present in the compound [1-4].
The history of this reagent goes back to 1975 when E J Corey and W Suggs suggested it as an oxidizing agent.
Preparation of PCC Reagent
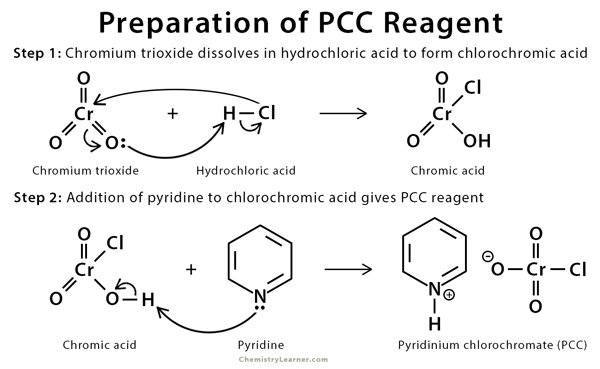
PCC reagent is prepared by adding 1 mol. of solid chromium trioxide to concentrated hydrochloric acid, followed by rapid stirring. The solution is cooled to 0 °C. To this solution, 1 mol. of pyridine is added until the final mixture turns into a yellow-orange solid. The solid product is filtered and dried in a vacuum [5].
PCC Reagent Reaction and Examples
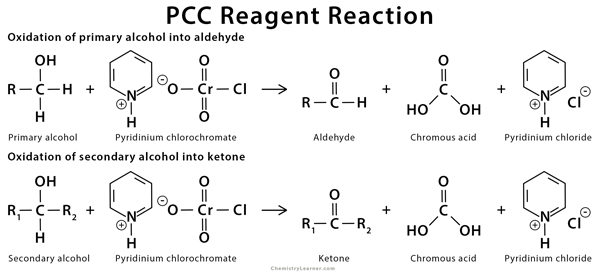
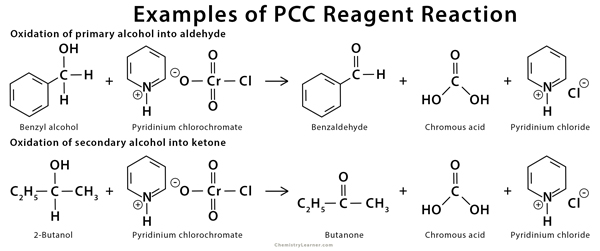
PCC reagent can oxidize both aliphatic and aromatic alcohol into an aldehyde [6-9].
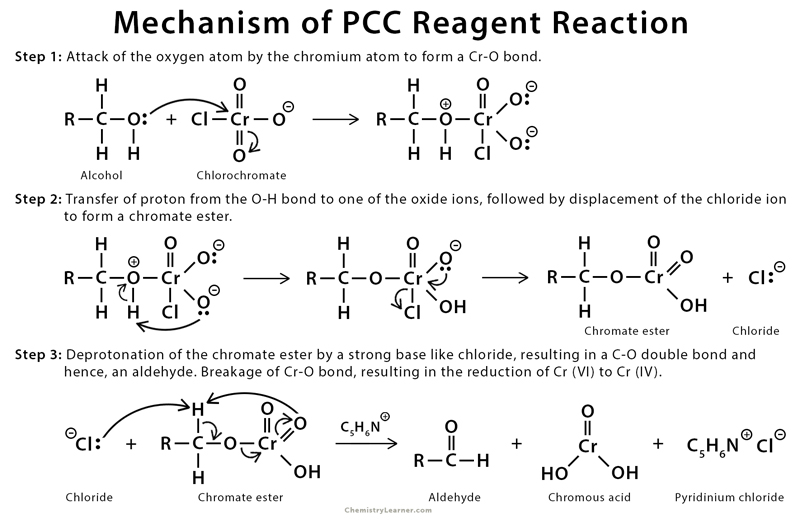
Mechanism of PCC Reagent Reaction
The alcohol coordinates with the chromium (IV) atom, displacing chlorine. The chlorine then acts as the base, resulting in oxidation of alcohol and reduction of chromium (VI) to chromium (IV).
FAQ
Ans. Jones’ reagent oxidizes primary alcohol into a carboxylic acid. In contrast, PCC reagent is very selective and oxidizes primary alcohol into aldehyde only.
References
- Definition – Chemistry.msu.edu
- Definition – Biologie.uni-hamburg.de
- Definition – Biologie.uni-hamburg.de
- Definition – Vanderbilt.edu
- Preparation – Nptel.ac.in
- Example – Ochempal.org
- Example – Chem.ucla.edu
- Example – Chem.libretexts.org
- Mechanism – Chem.libretexts.org