Perkin Reaction
Definition: What is Perkin Reaction?
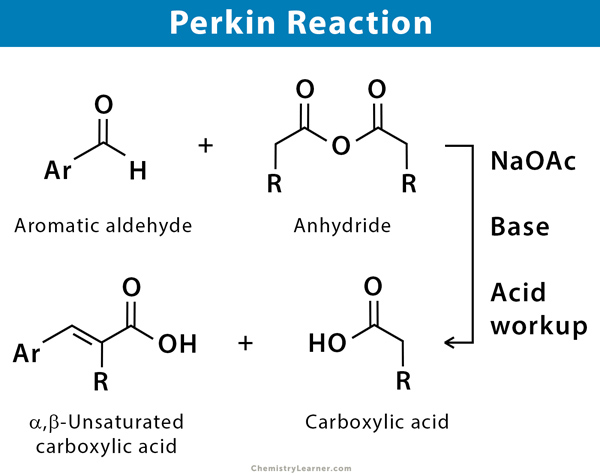
The Perkin reaction, also known as Perkin condensation, is an organic reaction which is used for the synthesis of α,β-unsaturated aromatic acid by the condensation of an aromatic aldehyde and an acid anhydride, in the presence of an alkali salt of the acid acting as a weak base. Generally, such type of reaction is only applicable to aromatic aldehyde and useful for the preparation of substituted cinnamic acid. One notable application for the Perkin reaction is in the laboratory synthesis of the phytoestrogenic stilbene resveratrol [1-4].
The reaction has been named after English chemist William Henry Perkin, who first reported in a journal in 1868.
Example of Perkin Reaction
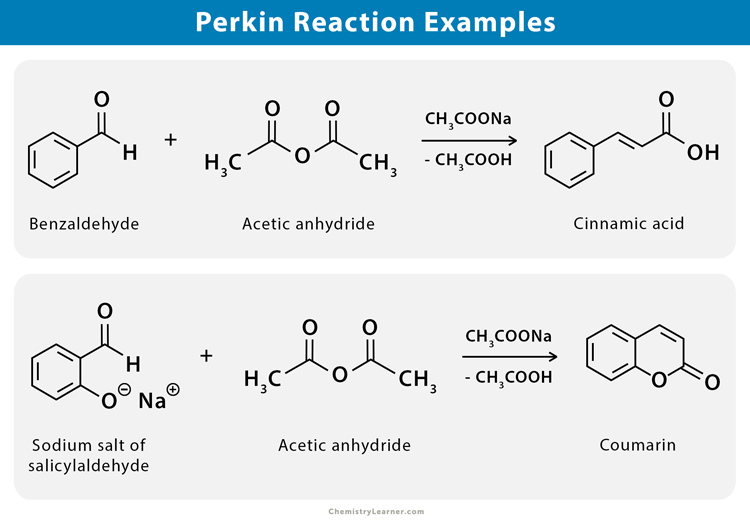
Perkin reaction is used to synthesize cinnamic acid from benzaldehyde. Aside, the synthesis of coumarin is carried out by heating the sodium salt of salicylaldehyde with acetic anhydride [1-3].
Mechanism of Perkin Reaction
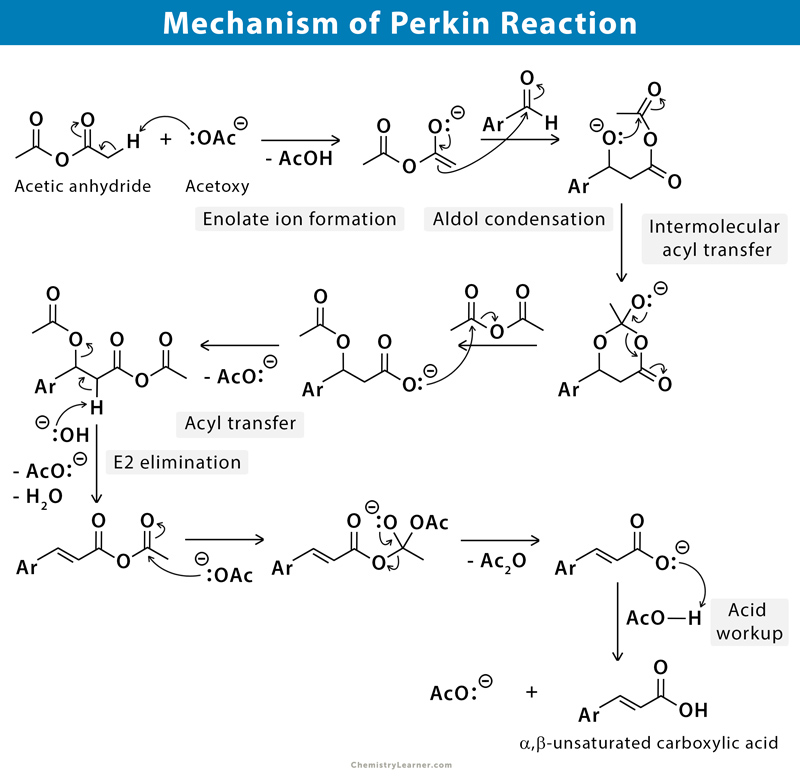
The mechanism goes through numerous steps, including several additions and eliminations. Formation of anhydride enolates and aldol type condensation provides the alkoxide anhydride. Intermolecular acylation generates an acetoxy carboxylate, which forms a mixed anhydride. On elimination of acetic acid and subsequent hydrolysis gives the unsaturated acid [1-3].
References
- Definition, example, and mechanism – Longdom.org
- Definition, example, and mechanism – Shodhganga.inflibnet.ac.in
- Definition, example, and mechanism – Researchgate.net
- Definition and mechanism – Chem.libretexts.org





Hey! I’m not still sure if you still monitor this page, but I think the structure of cinnamic acid is incorrect. I believe it should have an OH group instead of a H group.
Many thanks for pointing out the typo. We have corrected it.