pH Scale
pH is a measure of how acidic or basic a substance is. It takes a numerical value between 0 and 14 that measures the relative amount of free hydrogen and hydroxyl ions in water. Water having more hydrogen ion concentration than hydroxyl is acidic. On the other hand, water having more hydroxyl ion concentration than hydrogen is basic or alkaline [1-4].
In medical science, having the proper pH in the blood and other body fluids is essential for the body to function correctly. In agriculture, pH is used to determine the soil’s acidity and adjust the acidic content to make it suitable for growing specific crops.
How is pH Measured [1-5]
Formula
pH is measured on a logarithmic scale. The formula for pH is
pH = – log10 [H+]
Where [H+] is the hydrogen ion concentration.
Range and Indication
The range of pH goes from 0 to 14. A value less than 7 indicates that water is acidic. A value greater than 7 indicates that water is alkaline. A value equal to 7 shows that water is neutral. Each whole pH value below 7 is ten times more acidic than the next higher value. For example, a pH of 5 is ten times more acidic than 6. The same holds for pH values above 7. Each is ten times more basic than the next lower whole value. For example, a pH of 13 is ten times more basic than 12.
Device
The pH is usually measured with a pH meter, which refers to the difference in electromotive force (emf) between appropriate electrodes placed in the solution to be tested. These portable devices can be easily carried to the test site. Large devices for measuring pH are available in laboratories.
Examples of Acid and Alkaline Substances
Pure water has a pH of 7. When a chemical is added to it, the pH is altered. Examples of acidic substances are vinegar (acetic acid), lemon (citric acid), soda, and hydrochloric acid. When these are added to pure water, the pH drops below 7. Low pH values can corrode substances and dissolve metals [1-5].
Examples of alkaline substances are soap, detergents, baking soda, ammonia, and borax. When these substances are added to pure water, the pH value rises. A high pH value makes the taste bitter.
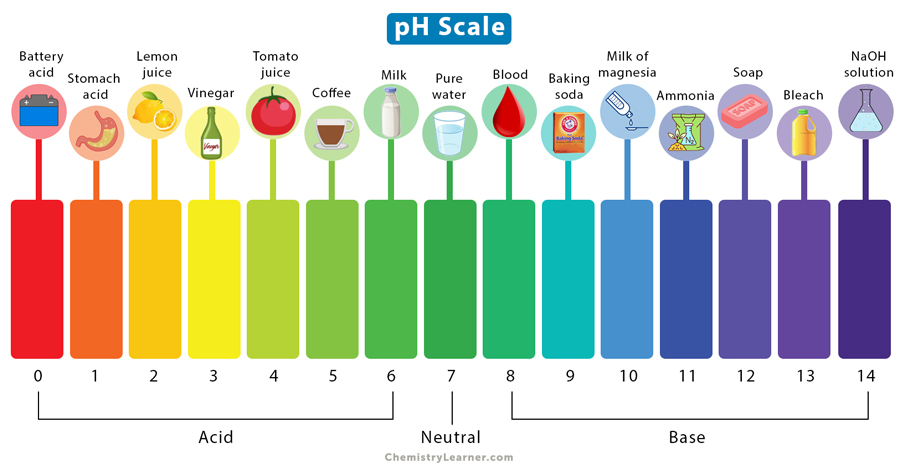
Examples of common substances, like food and chemicals, and their positions on the pH scale are shown below. The colors from green to red indicate an acidic solution, green to violet indicate a basic solution, and pure green indicates that a solution is neutral.
Importance of pH
pH is important because the presence of chemicals alters the acidity of water. Therefore, we can conclude whether water is acidic or basic by detecting the pH [1-5].
The pH of water determines a substance’s solubility and biological availability of nutrients such as phosphorus, nitrogen, and carbon. pH also indicates the amount of heavy metals such as iron, lead, and copper dissolved in water. For example, pH determines how much and what form of phosphorous can be utilized by aquatic life. The solubility of heavy metals determines their toxicity. Metals are more toxic at lower pH because they have high solubility.





it gud
Nice
Sir plz help me and better learn the chemistry.