Reductive Amination
Reductive amination is a fundamental and versatile chemical transformation essential in organic chemistry. This transformative process involves the conversion of carbonyl compounds, such as aldehydes and ketones, into primary and secondary amines by employing a reducing agent, typically in the presence of a catalyst. It is the basis for synthesizing various organic compounds, including pharmaceuticals, agrochemicals, and fine chemicals [1-3].
Examples
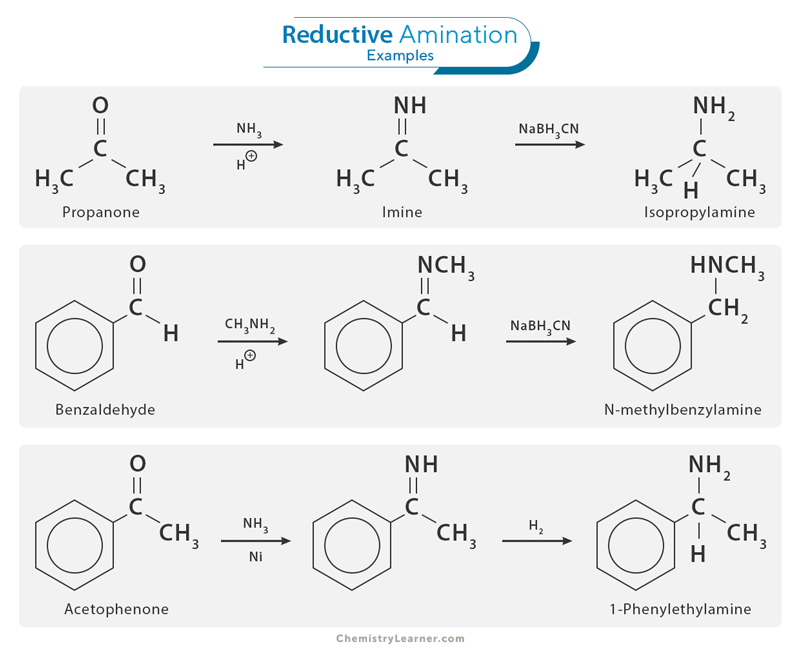
Here are a few examples of reductive amination [1,4,5].
1. Acetone to Isopropyl Amine
i. Acetone reacts with ammonia in the presence of an acid catalyst to form an imine.
CH3COCH3 + NH3 → CH3C(CH3)=NH2
ii. The imine is then reduced by sodium cyanoborohydride to yield a primary amine, isopropyl amine.
CH3C(CH3)=NH2 + NaBH3CN → CH3CH(CH3)-NH2
2. Benzaldehyde to N-methylbenzylamine
i. Benzaldehyde reacts with methylamine in the presence of acetic acid to form an imine.
C6H5CHO + CH3NH2 → C6H5CH=NCH3 + H2O
ii. The imine is then reduced by sodium cyanoborohydride in an acidic environment to yield a secondary amine, N-methylbenzylamine.
C6H5CH=NCH3 + NaBH3CN → C6H5CH2NHCH3
3. Acetophenone to 1-Phenylethylamine
i. Acetophenone reacts with ammonia in the presence of a nickel catalyst to form an imine with high stereoselectivity.
CH3COPh + NH3 → CH3C(Ph)=NH
ii. The imine is reduced to yield the chiral amine product, 1-phenylethylamine, with the desired stereochemistry.
CH3C(Ph)=NH + H2 → CH3CH(Ph)-NH2
Mechanism
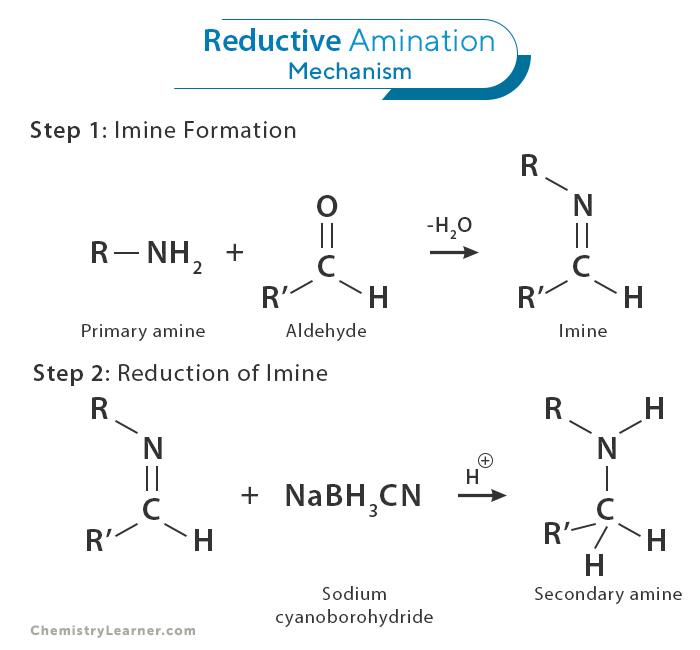
Reductive amination involves two steps to convert the carbonyl compound into an amine. Let us break down the critical steps in the mechanism [1,4]:
1. Formation of an Imine
The first step involves the reaction of the carbonyl compound with an amine, resulting in an imine. The imine is an intermediate compound that plays a pivotal role in the subsequent steps of the reaction. An acid or base typically catalyzes this step.
2. Reduction of the Imine
In this step, the imine is reduced to the desired amine. This reduction is achieved by introducing a reducing agent, which donates a hydrogen atom to the imine and breaks the double bond, leading to the amine formation. Typical reducing agents in reductive amination include metal hydrides like sodium borohydride (NaBH4) or sodium cyanoborohydride (NaBH3CN).
Role of Catalyst
Catalysts are often employed to enhance the efficiency and selectivity of the reductive amination process. Catalysts can help accelerate the reaction and control the stereochemistry of the resulting amine. Typical catalysts include transition metal complexes and organometallic compounds.
It is important to note that the mechanism of reductive amination can vary depending on the specific conditions, reagents, and catalysts used. Additionally, stereochemistry considerations are crucial, as the reaction can form chiral amines.
Applications
Some real-world applications highlighting the significance and impact of reductive amination in diverse fields are as follows [1]:
- Synthesis of many important antidepressant and antipsychotic medications
- Production of pharmaceutical intermediates and synthesis of chiral amines for the pharmaceutical industry
- Production of various agrochemicals and pesticides
- Responsible for scents and flavors in perfumes, cosmetics, and food products
- Development of advanced materials for various applications in aerospace and electronics