Saponification
What is Saponification
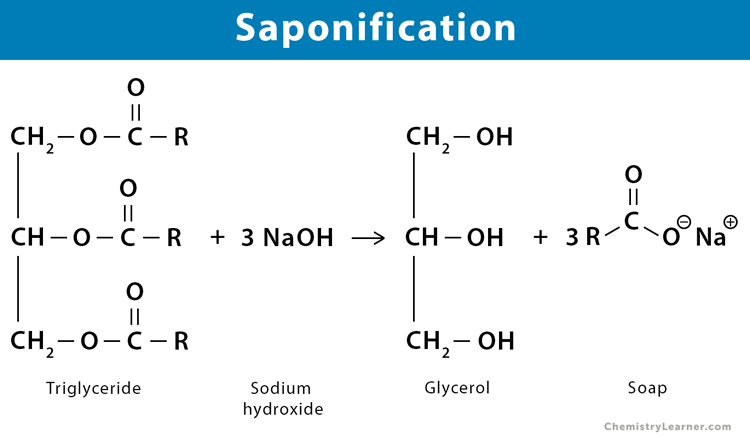
The process of converting fats, oil, and lipid into soap using an aqueous alkali is called saponification. Vegetable oil and animal fats are triesters or triglycerides that can be saponified in one or two steps. During this process, the triglyceride reacts with an aqueous hydroxide ion to form a mixture of glycerol and fatty acid derivative. The sodium or potassium salts of long-chained fatty acids are a significant component of soap [1-6].
General Equation for Saponification
During saponification, esters are cleaved in the presence of aqueous alkali to form an alcohol and an alkali-metal salt of carboxylic acid. The alkali used in this process is sodium hydroxide (NaOH) or lye for hard soap and potassium hydroxide (KOH) for soft soap.
The general reaction, using NaOH, is given by:
RCOOR’ + NaOH → ROH + R’COO– Na+
Here is a simple video explaining the saponification process:
Examples of Saponification [2-6]
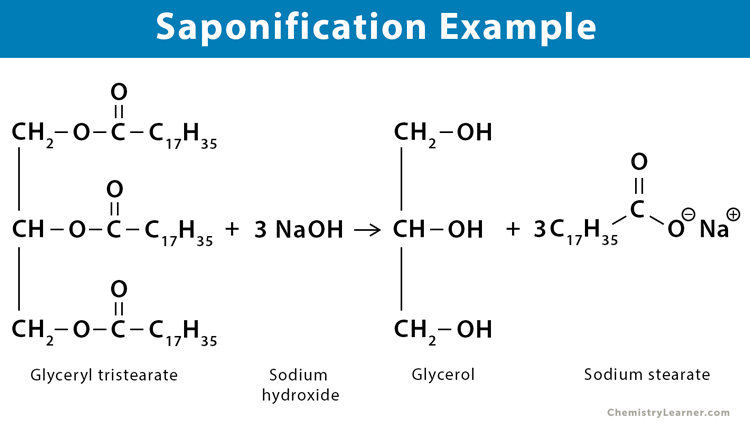
1. Sodium Stearate
Sodium stearate (C18H35NaO2) is the sodium salt of stearic acid (C18H36O2). It is used as a soap and a detergent. It is a significant component of soaps and contains both a hydrophobic and a hydrophilic part. It is produced when glyceryl tristearate ((C18H35O2)3C3H5), the triglyceride of stearic acid, is hydrolyzed with aqueous sodium hydroxide (NaOH).
(C18H35O2)3C3H5 + 3 NaOH → C3H5(OH)3 + 3 C18H35O2Na
2. Sodium Palmitate
Sodium palmitate (C16H31NaO2) is the sodium salt of fatty palmitic acid (C16H32O2). It is found in soaps and detergents. It can be derived by the saponification of glyceryl palmitate ((C16H31O2)3C3H5) using sodium hydroxide (NaOH) in the form of caustic soda, lye, or lime.
(C16H31O2)3C3H5 + 3 NaOH → C3H5(OH)3 + 3 C16H31O2Na
3. Methyl Salicylate
Methyl salicylate (HOC6H4COOCH3) reacts with sodium hydroxide (NaOH) to form a thick white solid of sodium salicylate (HOC6H4COO–Na+).
HOC6H4COOCH3 + NaOH → HOC6H4COO–Na+ + CH3OH
4. Methyl Acetate
Methyl acetate (CH3COOCH3) saponifies in the presence of sodium hydroxide (NaOH) to sodium acetate (CH3COO−Na+).
CH3COOCH3 + NaOH → CH3COO−Na+ + CH3OH.
5. Methyl Benzoate
Methyl benzoate (C8H8O2) reacts with aqueous sodium hydroxide (NaOH) to give sodium benzoate (water-soluble) and methanol (miscible with water).
C8H8O2 + NaOH → C7H5O2Na + CH3OH
Saponification Mechanism
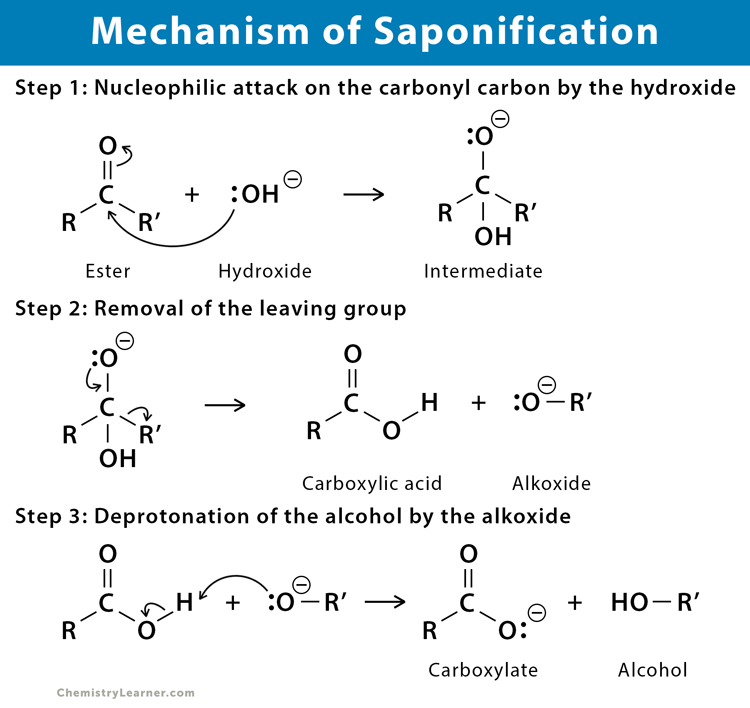
The mechanism of saponification is a nucleophilic carbonyl substitution process, which is explained in the following steps [4-6].
Step 1: The nucleophilic hydroxide ion attacks the ester group and forms an intermediate.
Step 2: The intermediate splits by releasing the leaving group to form a carboxylic acid and an alkoxide.
Step 3: Deprotonation removes the hydrogen from the carboxylic acid resulting in a carboxylate ion and alcohol.
Saponification Value
Saponification value (SV) or saponification number (SN) represents the number of milligrams of potassium hydroxide (KOH) required to saponify one gram of fat under a specific condition. The saponification value is a significant parameter used to characterize and evaluate the quality of edible fats and oils. Also, the saponification number provides information about the fatty acids’ average molecular weight. The higher the number, the lower the molecular weight of all fatty acids [7].
Unit: mg KOH/g
The following table gives the saponification values of various oils and fats.
| Oil/ fat | Saponification value (mg KOH/ g) |
|---|---|
| Canola oil | 182 – 193 |
| Sunflower oil | 189 – 195 |
| Olive oil | 184 – 196 |
| Soyabean oil | 187 – 195 |
| Coconut oil | 248 – 265 |
| Cottonseed oil | 189 – 207 |
| Palm kernel oil | 230 – 254 |
| Palm oil | 190 – 209 |
| Castor oil | 176 – 187 |
Applications of Saponification
The main application of saponification is in the manufacture of soaps. Different kinds of soaps serve different purposes like laundry, cleaning, and lubrication. Soaps may be precipitated by salting them out with saturated sodium chloride. Saponification also works with fire extinguishers. Fire extinguishers use it to convert burning fats and oil into non-combustible soap, which helps to reduce fire[7].
Saponification is significant in the food industry because it helps to know the amount of free fatty acid in a food item. The amount of free fatty acid can be distinguished by determining the quantity of alkali added to the fat or oil to make it neutral.
Esterification vs. Saponification[8]
Why is Saponification the Reverse of Esterification
During esterification, acid and alcohol combine to form an ester and release water. On the other hand, saponification breaks the bonds in an ester to form alcohol and long-chained fatty acid derivative.
Example of Esterification
C2H5OH + CH3COOH → CH3COOC2H5OH + H2O
The following table shows the difference between saponification and esterification.
| Esterification | Saponification | |
|---|---|---|
| Definition | Process of preparing ester | Process of preparing soap |
| Process | Acid reacts with alcohol in the presence of concentrated sulfuric acid | Ester reacts with aqueous alkali |
| Heat | Yes | No |
| Catalyst | Acid | Base |
| Byproduct | Water | Alcohol |
FAQs
Ans. Ethanol is less polar than water. It helps to dissolve the nonpolar fat so that it can react with sodium hydroxide.
Ans. The saponification reaction is endothermic as it takes heat from the surroundings.





Wow the page is so good
Wonderful illustration indeed.may you include some essential precautions for the reaction, possible dangers to one performing the experiment/s or procedures, necessary condition to be maintained.
Also attached a model video for the process and designed small plant to perform the procedure.
Thank you for your suggestion. We have included a simple video explaining the process. However, the essential precaution, possible dangers, and designing a small plant are beyond the scope of the article.
actually but i think to avoid all such of risk better put on your ppe during the process
I love knowing the chemical reaction. I am on batch four and switched from well water (calcium carbonate and ferrous something) to distilled water and wow what a difference. I am a retired engineer not a chemist but I wonder what the reaction would be.