Schiff Reagent
Definition: What is Schiff Reagent?
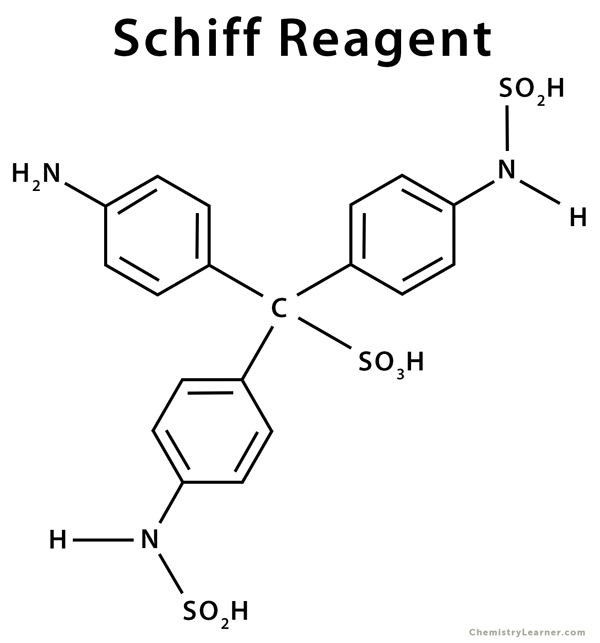
Schiff reagent is the product formed in some dye formulation reactions, such as the reaction between sodium bisulfite and fuchsine. It is used to check for the presence of aldehyde in an analyte. It can be used to differentiate between aldehydes and ketones. Fuschsine or rosaniline hydrochloride is a magenta-colored dye with a chemical formula C20H20N3·HCl, which is decolorized by sodium bisulfate [1,2].
Composition of Schiff Reagent [3]
Schiff reagent has a chemical formula given by C19H21N3S2O7·4H2O and is composed of:
Basic Fuchsin Hydrochloride (C20H19N3·HCl): <1%
Hydrochloric Acid (HCl): <1%
Sodium Metabisulfite (Na2S2O5): <1%
Water (H2O): >98%
Preparation of Schiff Reagent [4]
- Dissolve 5 g of basic fuchsine in 900 ml of boiling distilled water
- Cool to approximately 50 °C and slowly add 100 ml of 1M HCl
- Cool to approximately 25 °C and dissolve 10 g of Na2S2O5
- Shake for 3 minutes and incubate in the dark at room temperature for 24 hours
- Add 5 g of fine activated charcoal and shake for 3 minutes
- Filter solution (should be clear)
- Store at 4 °C in a foil-covered bottle
Schiff Reagent Test
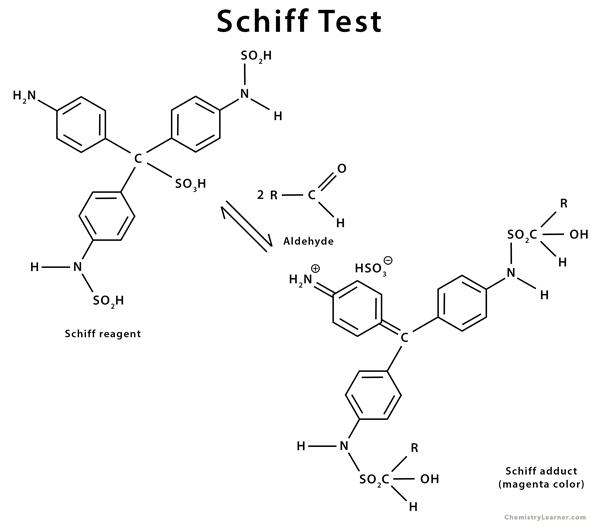
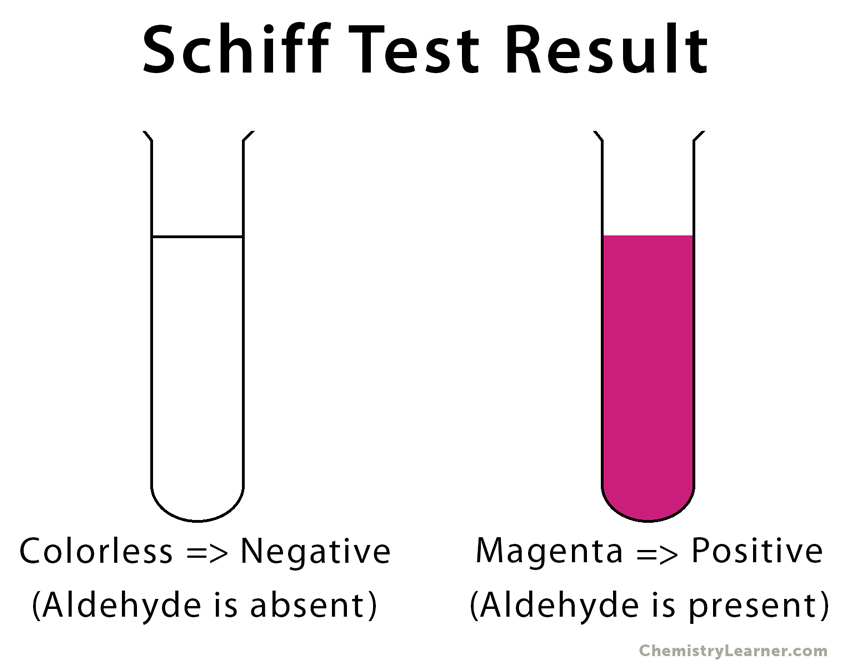
When an unknown solution containing aldehyde is added to the colorless Schiff reagent, it reverts to its original magenta color. This test is known as the Schiff test [5,6].
The history of the Schiff test goes back to 1866 when German-Italian chemist Hugo Schiff developed it and reported it in a journal.
FAQs
Ans. In its open structure, glucose contains an aldehyde group but forms a cyclic acetal structure(also called pyranose). This structure is quite stable and is not broken down by Schiff’s reagent, which is a weak base. Hence, it does not form hydrazone giving a negative Schiff’s test.





Cool to approximately 25 °C and dissolve 10 g of K2S2O5..
K2 or Na2 ????
Thank you for your comment. It is Na2 and we have made the correction.
This one of the most incredible site I have browsed on so far,as a medical laboratory science student, this site has helped me a lot.
Thanks for your assistance.