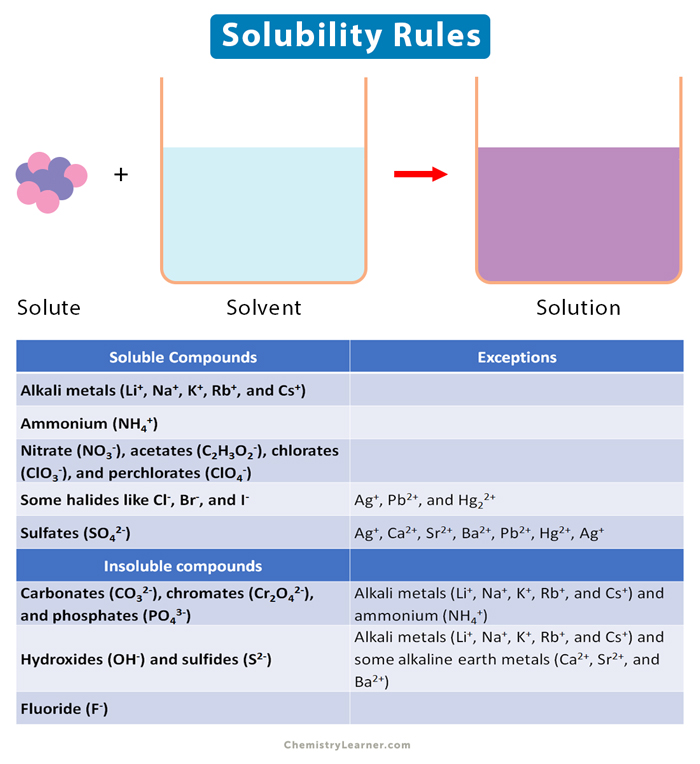
Solubility Rules
When solute mixes with solvent, it can dissolve and form a solution or fall to the bottom as a precipitate. Our article on solubility explains how ionic compounds dissolve in water to form a solution. However, to predict whether the compound will dissolve or not, we need to know a few general rules. This set of rules is collectively known as solubility rules. They are specifically used to predict the solubility of ionic salts in water. They are essential for many laboratory processes and for preparing medicine [1-4].
List of Solubility Rules [1-4]
- Salts of alkali metals are soluble. These include lithium (Li+), sodium (Na+), potassium (K+), rubidium (Rb+), and cesium (Cs+).
- Ammonium (NH4+) salts are soluble.
- Salts of nitrate (NO3–) are usually soluble. So are acetates (C2H3O2–), chlorates (ClO3–), and perchlorates (ClO4–).
- Salts of some halides are generally soluble. These include chloride (Cl–), bromide (Br–), and iodide (I–). Some exceptions exist. The salts of silver (Ag+), lead (Pb2+), and mercury (Hg22+), like AgCl, PbBr2, and Hg2Cl2, are insoluble.
- Most silver (Ag+) salts are not soluble. The exceptions include silver nitrate (AgNO3) and silver acetate Ag(C2H3O2).
- Most sulfate (SO42-) salts are soluble. However, there are some exceptions. These include calcium sulfate (CaSO4), strontium sulfate (SrSO4), and barium sulfate (BaSO4). Lead sulfate (PbSO4), silver sulfate (Ag2SO4), and mercuric sulfate (HgSO4) are slightly soluble.
- Most hydroxide (OH–) salts are sparingly soluble. The salts of alkali metals are soluble. The hydroxides of alkaline earth metals, like magnesium (Mg2+), calcium (Ca2+), strontium (Sr2+), and barium (Ba2+), are mildly soluble. The hydroxides of transition metals and aluminum (Al3+) are insoluble – like cobalt hydroxide Co(OH)2, ferric hydroxide Fe(OH), and aluminum hydroxide Al(OH)3.
- Most sulfides (S2-) of transition metals are extremely insoluble. These include cadmium sulfide (CdS), iron sulfide (FeS), zinc sulfide (ZnS), silver sulfide (Ag2S), and lead sulfide (Pb2S)
- Carbonates (CO32-) usually are insoluble. Carbonate of alkaline earth metals, like calcium carbonate (CaCO3), strontium carbonate (SrCO3), and barium carbonate (BaCO3), along with ferrous carbonate Fe(CO3) and lead carbonate (PbCO3), are insoluble.
- Chromates (Cr2O42-) are often insoluble. These include lead chromate (PbCr2O4) and barium chromate (BaCr2O4).
- Phosphates (PO43-) are frequently insoluble. These include calcium phosphate (Ca3(PO4)2) and silver phosphate (Ag3(PO4)2).
- Fluorides (F–) are commonly insoluble. These include magnesium fluoride (MgF2), barium fluoride (BaF2), and lead fluoride (PbF2).
The following table summarizes the solubility rules.
| Soluble Compounds | Exceptions |
|---|---|
| Alkali metals (Li+, Na+, K+, Rb+, and Cs+) | |
| Ammonium (NH4+) | |
| Nitrate (NO3–), acetates (C2H3O2–), chlorates (ClO3–), and perchlorates (ClO4–) | |
| Some halides like Cl–, Br–, and I– | Ag+, Pb2+, and Hg22+ |
| Sulfates (SO42-) | Ag+, Ca2+, Sr2+, Ba2+, Pb2+, Hg2+, Ag+ |
| Insoluble compounds | |
| Carbonates (CO32-), chromates (Cr2O42-), and phosphates (PO43-) | Alkali metals (Li+, Na+, K+, Rb+, and Cs+) and ammonium (NH4+) |
| Hydroxides (OH–) and sulfides (S2-) | Alkali metals (Li+, Na+, K+, Rb+, and Cs+) and some alkaline earth metals (Ca2+, Sr2+, and Ba2+) |
| Fluoride (F–) |
FAQs
Ans. AgCl has a very low solubility of 0.002 g/L. It is insoluble in water and precipitates out.
Ans. PbCl2 is poorly soluble in cold water. However, it goes into a solution in warm water.
Ans. Cobalt chloride is soluble in water and forms a red solution.
Ans. Ethanol is soluble in water since it is a polar compound. It has an OH group that primarily forms a hydrogen bond with water.
Ans. Calcium carbonate has a very low solubility (15 mg/L at 25 ˚C in water).
Ans. Barium sulfate is insoluble in water at room temperature.
Ans. Water does not dissolve in hexane because it cannot form hydrogen bonds.
Ans. No calcium oxalate or CaC2O4 is not soluble in water.
Ans. Manganese (III) acetate is moderately soluble in water.
Ans. PbS displays a very low solubility in water.