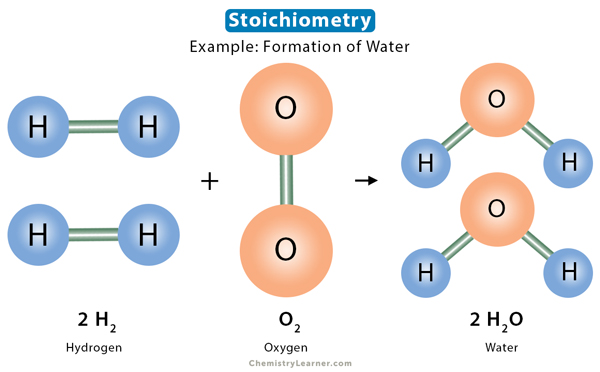
Stoichiometry
Stoichiometry is the study of the relationship between the quantity of reactants and products in a chemical reaction. German chemist Jeremias Benjamin Richter was the first to define stoichiometry in 1792 [1-4].
Theory
Stoichiometry is based on the principle of the law of conservation of mass. According to this law, the total mass of the reactants is equal to that of the product. Since chemical reactions neither create nor destroy matter, the amount of each element is the same throughout the reaction. The number of atoms of a particular element on the reactant side is equal to the number of atoms of that element on the product side [1,2].
The relationship among the quantities of the reactants and products is a positive integral ratio. If the amounts of each reactant are known, each product’s quantity can be easily determined. In this context, we refer to a stoichiometric amount of a reactant, which is an optimum amount where the following happens:
- The reaction proceeds to a completion
- All the reactants are consumed
- There is no deficiency of reagents
- There is no excess of reagents
Balancing Chemical Equations
A chemical equation represents a chemical reaction with reactants on the left side and products on the right side. Numbers are placed before each reactant and product to balance the reaction [1-5].
To illustrate this concept, let us take the following example of a chemical reaction. Ethanol (C2H5OH) reacts with oxygen (O2) to produce carbon dioxide (CO2) and water (H2O).
C2H5OH + O2 → CO2 + H2O
The equation is not balanced. Let us proceed to balance the equation.
First, we must ensure that the number of carbon atoms is equal on both sides. We multiply CO2 by 2.
C2H5OH + O2 → 2 CO2 + H2O
Next, we ensure that the number of hydrogen atoms is equal. We multiply H2O by 3.
C2H5OH + O2 → 2 CO2 + 3 H2O
Finally, we balance the oxygen by multiplying O2 by 3.
C2H5OH + 3 O2 → 2 CO2 + 3 H2O
Therefore, 1 mole of C2H5OH reacts with 3 moles of O2 to produce 2 moles of CO2 and 3 moles of H2O.
Stoichiometric Coefficients
Stoichiometric coefficients are the numbers before an atom, ion, or molecule in a chemical reaction. They are essential because they establish the mole ratios between the reactants and products. In the above example, the mole ratio between C2H5OH and CO2 is 1:2, and that between C2H5OH and H2O is 1:3. Therefore, if we know the number of moles of C2H5OH present in the reaction, we can quickly determine the quantities of CO2 and H2O produced in the reaction.
So far, we have discussed quantifying a chemical equation in moles. We can easily convert from moles to mass in grams by multiplying it with the molecular weight of the substance.
Converting Moles to Grams
Suppose 0.5 moles of C2H5OH is present in the above reaction. The molar mass of C2H5OH is = 2 x 12 g + 6 x 1 g + 16 g = 46 g. The quantity of C2H5OH in grams is = 0.5 M x 46 g M-1 = 23 g.
Converting Grams to Moles
Suppose 50 g of CO2 is produced in the reaction. The molar mass of CO2 is = 12 g + 2 x 16 g = 44 g. The number of moles of CO2 produced is = 50 g / 44 g M-1 = 1.14 M.
The mole ratio can convert the amounts of any two substances in a chemical reaction from moles to grams and vice versa. Let us look at the solved problems below to understand this concept.
Example Problems
Problem 1. According to the following chemical equation, how many moles of nitrogen (N2) must be used to produce 5 moles of ammonia (NH3)?
N2 + 3 H2 → 2 NH3
Solution
2 moles of NH3 can be produced by 1 mole of N2. Therefore, 5 moles of NH3 can be produced by
(½) x 5 = 2.5 moles of N2.
Problem 2. Propane (C3H8) burns in the presence of oxygen as follows:
C3H8 + 5 O2 → 4 H2O + 3 CO2
If 100 g of propane is burned, how many grams of carbon dioxide (CO2) is produced?
Solution
Molar mass of propane (C3H8) = 3 x 12 g + 8 x 1 g = 44 g
Number of moles in 100 g of propane = 100 g/ 44 g M-1 = 2.27 M
1 mole of propane produces 3 moles of CO2. Therefore, 2.27 M of propane produces 2.27 x 3 = 6.82 M of CO2.
Molar mass of CO2 = 12 g + 2 x 16 g = 44 g
6.82 moles of CO2 = 6.82 M x 44 g M-1 = 300 g