Suzuki Reaction
Definition: What is Suzuki Reaction?
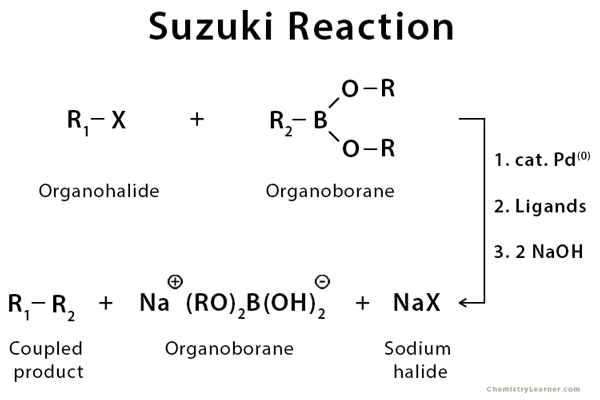
A coupling reaction in organic chemistry is a general term for a variety of reactions where two fragments are joined together with the aid of a metal catalyst. The Suzuki reaction, or Suzuki-Miyaura coupling reaction, is an organic coupling reaction of an organohalide with an organoborane to give a coupled product using a palladium catalyst and base. Boronic acid or boronic ester, or exceptional cases of aryl boron trifluoride can be used as well. This reaction is used to create carbon-carbon bonds to produce conjugated systems of alkenes, styrenes, or biaryl compounds [1-5].
The history of this reaction goes back to 1979 when Japanese Nobel Laurate Akira Suzuki first published this work in a scientific journal.
Examples of Suzuki Reaction
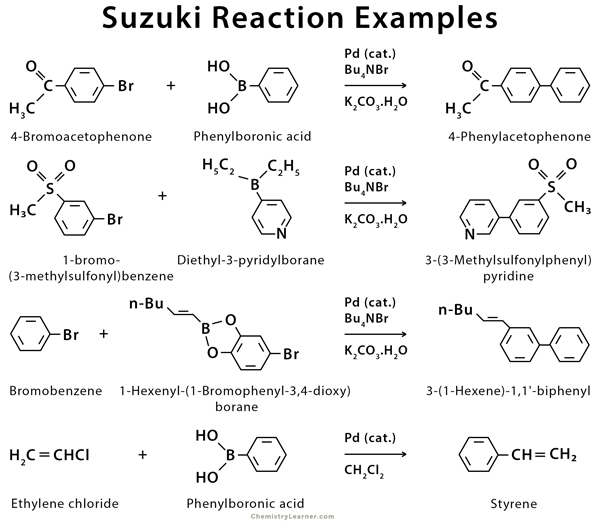
Examples of Suzuki reaction include aryl, alkenyl (vinyl), and alkynyl halides reacting with aryl, alkenyl (vinyl), or alkynyl organoborane [2-7].
Mechanism of Suzuki Reaction
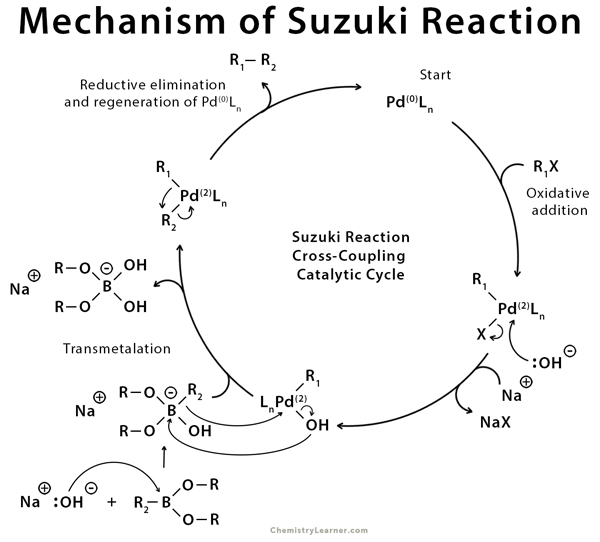
The Suzuki coupling mechanism follows a catalytic cycle involving three primary steps. These are oxidative addition, transmetalation, and reductive elimination. The first step is the oxidative addition of aryl halides to the Pd(0) complex to give intermediate 1, a Pd(2) species. Under the action of a base, an organoborane compound reacts with intermediate 1 in transmetalation to give intermediate 2. This step is followed by reductive elimination to give the desired product and regenerate the original Pd(0) species [2-8].
Applications of Suzuki Reaction
Suzuki reaction can conjoin a variety of aryl halides and alkenyl halides with alkenylboranes and arylboronic acids and is thus an essential method of synthesizing many styrenes, alkenes, and biphenyls. It is very cost-effective for use in the synthesis of intermediates for pharmaceuticals or fine chemicals.
References
- Definition and mechanism – Chem.libretexts.org
- Definition, example, and mechanism – Name-reaction.com
- Definition, example, and mechanism – Ocf.berkeley.edu
- Definition, example, and mechanism – Mdpi.com
- Definition, example, and mechanism – Pubs.rsc.org
- Example and mechanism – Organic-chemistry.org
- Example and mechanism – Harvard.edu
- Mechanism – Chemtube3d