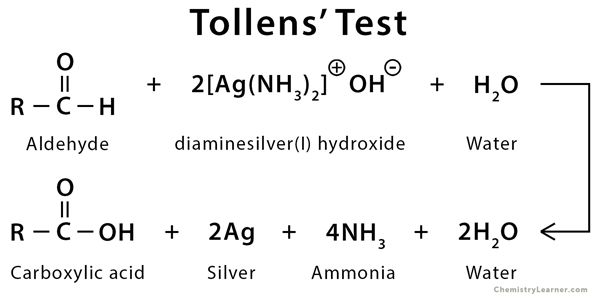
Tollens’ Test
Definition: What is Tollens’ Test?
Tollens’ test, also known as a silver-mirror test, is a qualitative laboratory test that is used to differentiate between an aldehyde and a ketone. It uses the fact that aldehydes can be more easily oxidized than ketones. This test cannot distinguish between an aldehyde and an α-hydroxy ketone (e.g., hydroxy acetone) [1-4].
During the process, two events occur. First, the aldehyde gets converted into carboxylic acid. Second, the Ag+ ions are reduced to Ag metal, which leaves a shiny appearance inside the test tube, indicating a positive result.
The history of this reaction goes back to 1882 when a work done by German chemist Bernard Tollens was published in a German journal.
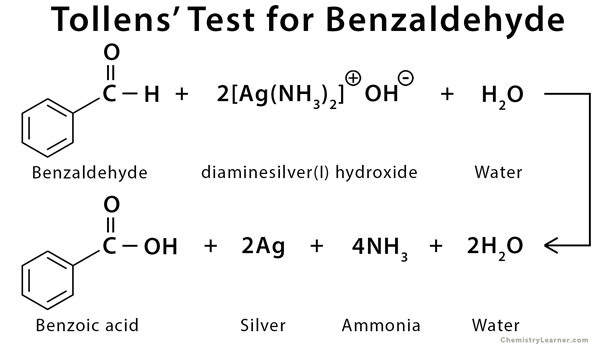
Examples of Tollens’ Test
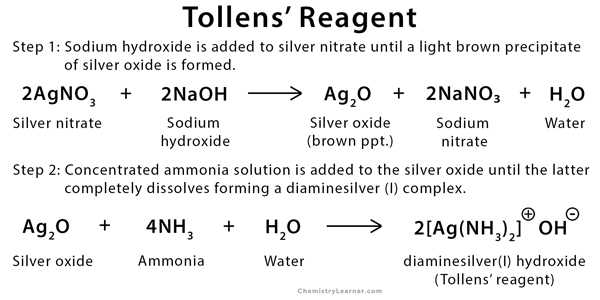
Tollens’ Reagent
Tollens reagent is a mild oxidizing chemical reagent that is used in the Tollens’ test. It is a colorless, basic, and aqueous solution containing silver ions coordinated to ammonia, forming a diamine silver(I) complex [Ag(NH3)2]+. Tollens’ reagent is prepared using a two-step procedure. [1-3]
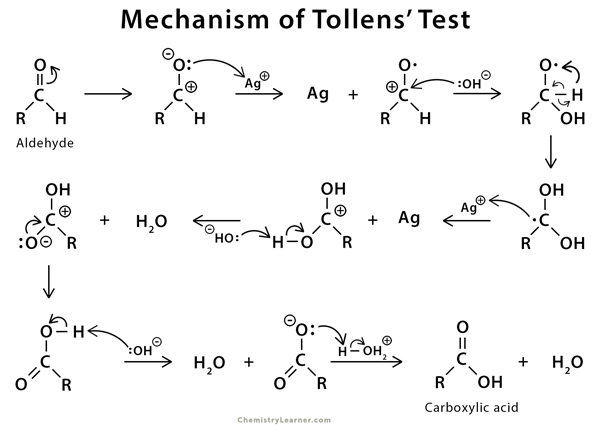
Mechanism of Tollens Test [5]
FAQs
Q.1. Why does fructose reduce Tollens’ reagent?
Ans. In an aqueous solution, fructose is enolized and converted into aldehyde in a basic medium. As aldehydes generally reduce Tollen’s reagent, thus fructose also reduces Tollen’s reagent.
References
- Definition and preparation – Chem.libretexts.org
- Definition and preparation – Byjus.com
- Definition and preparation – Vedantu.com
- Definition – Edu.rsc.org
- Mechanism – Leah4sci.com





So much helpful…
What is the importance of the Tollen Test, Can anyone give me 10 reasons of it?
As mentioned in the article, the test is used to differentiate between an aldehyde and a ketone.
Hello, [Ag(NH3)2]+ and OH- are forming an ionic pair?
I assume that the reaction would not occur in an acid medium or at pH 7?
Thanks!
They exist as ions in the solution. The medium is basic. It cannot be acidic.