Valence Bond Theory
Several theories effectively explain the electronic configuration of atoms. Quantum mechanics allows us to predict specific regions surrounding an atom where electrons are observed to congregate. For example, s orbitals adopt a spherical form, p orbitals adopt a dumbbell shape, and so on. Nonetheless, these projections exclusively outline the orbitals encircling solitary atoms. In molecular formation through atom bonding, atomic orbitals cannot explain electron distribution within the molecule. A more comprehensive study of electron allocation is necessary to understand the electronic arrangement of molecules. One prominent theory suggests that a covalent bond occurs when a pair of electrons is jointly shared by two atoms, simultaneously attracted by both the atoms’ nuclei. [1-4]
Valence bond theory is based on the idea that a covalent bond is a consequence of the convergence of half-filled atomic orbitals, each harboring a lone electron, which culminates in sharing an electron pair between the two conjoined atoms. According to valence bond theory, a covalent bond forms when two criteria are met: (1) an orbital on one atom overlaps with an orbital on a different atom, and (2) the unpaired electrons of each orbital merge, generating an electron pair. The mutual attraction between this negatively charged electron pair and the positively charged nuclei of the two atoms leads to the physical association of the atoms through a binding force resulting in a covalent bond. The strength of a covalent bond lies in the degree of orbital overlap. Extensively overlapping orbitals give rise to stronger bonds than those characterized by marginal overlap.
Postulates
Below are the fundamental principles of the valence bond theory: [1-4]
- Covalent bonding emerges when two valence orbitals, each half-filled, from distinct atoms coincide. This overlapping increases the electron density within the intermediate space between the bonded atoms, enhancing the molecule’s stability.
- Numerous unpaired electrons within an atom’s valence shell result in multiple bonds with other atoms. On the contrary, paired electrons within the valence shell do not participate in chemical bonds.
- The directionality of covalent chemical bonds is such that it aligns with the spatial orientation of overlapping atomic orbitals. This alignment ensures that bonds are formed parallel to the region covered by the corresponding atomic orbitals.
- Characteristic patterns of atomic orbital overlap differentiate sigma bonds from pi bonds. Pi bonds result from lateral overlap, while sigma bonds form through overlap along the axis containing the nuclei of the two atoms.
Examples [1-3]
Hydrogen (H2)
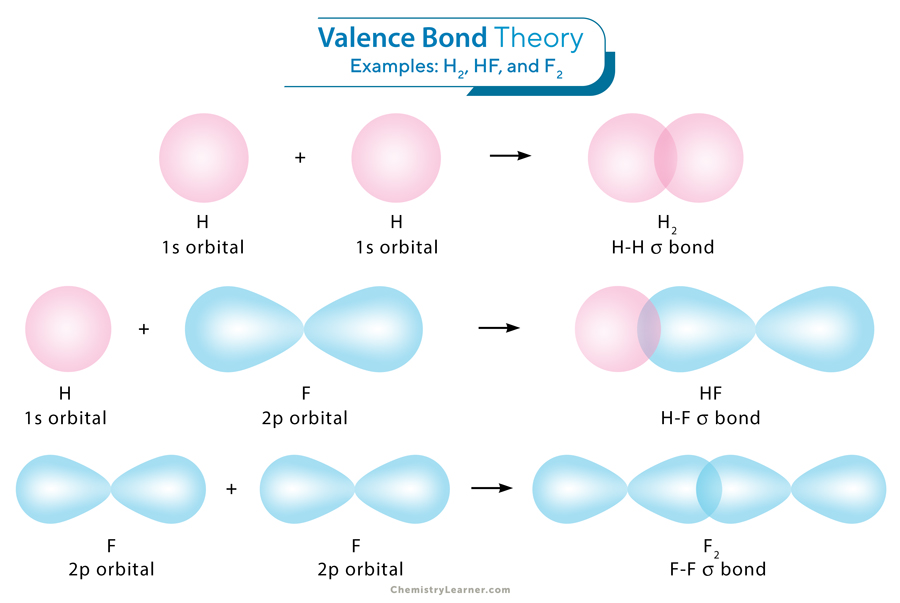
Let us consider the simple case of the hydrogen molecule H2. In a hydrogen atom, the electronic configuration is 1s1, signifying the presence of one valence electron within the spherical 1s orbital.
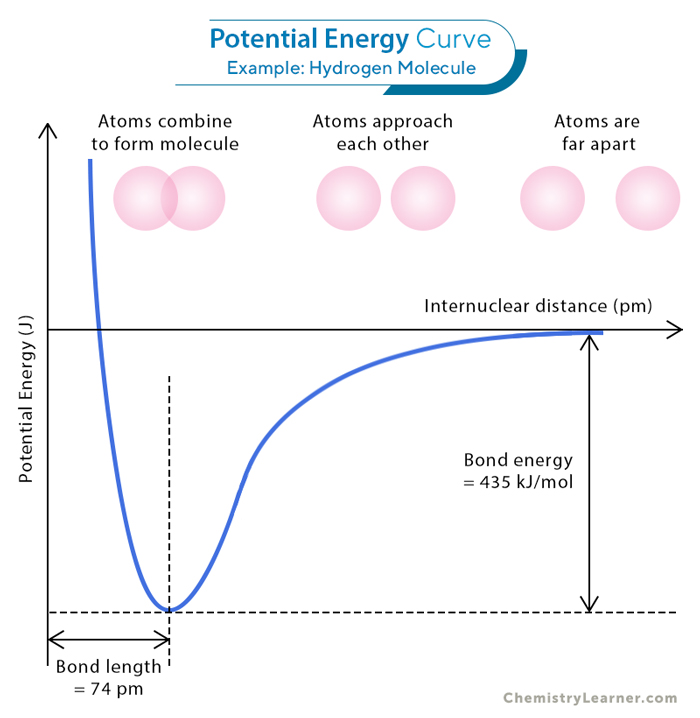
As two hydrogen atoms draw closer, their respective 1s orbital intermingle, and the two 1s electrons pair and form overlapping orbitals. These shared electrons encounter the simultaneous attraction of both hydrogen nuclei, effectively binding the two nuclei together. The energy diagram below summarizes the molecule’s potential energy versus the distance between the two hydrogen nuclei.
In the initial state, where the two atoms are far apart, no overlap or interaction between them occurs. However, their respective orbitals overlap as they draw nearer, prompting an attraction between one atom’s nucleus and the other’s electron. This interaction leads to a reduction in the potential energy. This energy minimizes when the atoms achieve an optimal distance, known as the bond length. In the case of the H2 molecule, the bond length is 74 picometers, often denoted as 0.74 Å (1 Å = 10-10 meters).
The difference in energy between the most stable state, characterized by the lowest energy at the optimum distance, and the state where the atoms are entirely separate is called bond energy or bond dissociation energy. This bond energy equals 7.22 × 10-19 J or 435 kJ/mol for a single H-H bond. However, as the atoms approach each other closer than the optimal distance, the repulsion originating from the nuclei’s proximity takes priority, elevating the molecule’s energy further.
Another crucial feature of the covalent bond in H2 is how the two 1s orbitals interact; that is, they align head-to-head. This specific type of overlap gives rise to a sigma (σ) bond. The 1s orbital is characterized by spherical symmetry. Therefore, the σ bond forms circles when cross-sectional planes are taken across the bond at any point.
Hydrogen Fluoride (HF)
The valence bond theory can also explain hydrogen fluoride (HF) bonding. Here, the 1s orbital in hydrogen and the 2p orbital of fluorine overlap. The fluorine atom has the valence electron configuration of 2s2 2p5. The 2s has a filled orbital; the 2p has two filled orbitals with one half-filled. The half-filled orbital has one single electron. The filled orbital cannot form bonds, so only the half-filled 2p is available for overlap. Therefore, hydrogen’s 1s orbital overlaps head-to-head with fluorine’s half-filled 2p orbital to form the H-F σ bond.
Fluorine (F2)
A σ bond can also arise from the overlap of two p orbitals. In the case of molecular fluorine (F2), a σ bond forms through the convergence of two half-filled 2p orbitals, one arising from each fluorine atom.
For molecules containing double or triple bonds, one of these bonds is a sigma bond, and the others are a different type of bond known as a pi bond (π bond). Pi bonds result from the sideways overlap of p orbitals, resulting in high electron density on opposite sides of the internuclear axis.
Limitations
The valence bond theory has limitations, especially when dealing with organic molecules such as CH4. The valence electron configuration of a carbon atom is 2s²2p². The 2s orbital is completely filled, while the 2p orbitals have two half-filled and one unoccupied [1].
According to the valence bond theory, given the presence of two half-filled orbitals, the carbon atom should be capable of forming two bonds. However, carbon consistently forms four bonds in any stable organic compound. Therefore, we need to address the bonding of carbon and other atoms that defy the straightforward application of the valence bond theory. To accomplish this, a supplementary theory known as orbital hybridization is introduced. This theory complements the valence bond theory in explaining these situations.
FAQs
Ans. The critical difference is that the molecular orbital theory describes the molecular orbital formation. In contrast, the valence bond theory describes atomic orbital formation.