Addition Reaction
Definition: What is Addition Reaction?
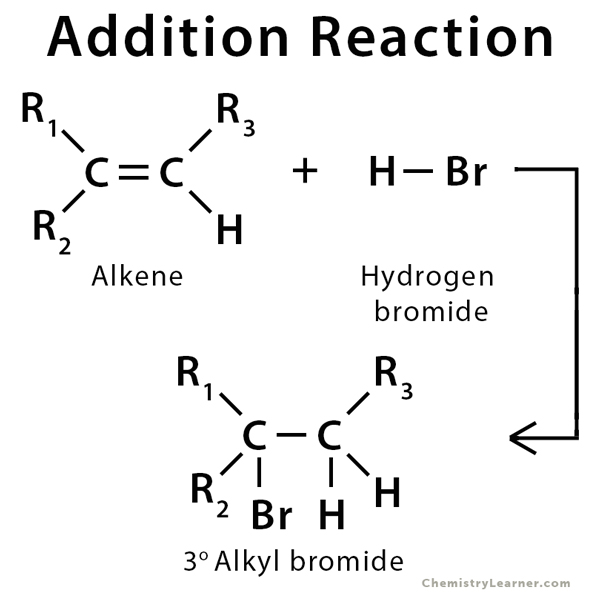
Addition reaction occurs when two or more reactants combine to form a product without the loss of any atoms present in the reactants. Addition reaction is common in compounds that have unsaturated C-C bond, like double (alkene) and triple (alkyne) bonds. The weaker π bond is converted into two new stronger σ bonds. Alkenes can give rise to a wide variety of products with various regioselectivity and stereoselectivity depending on the reagent and reaction conditions. There are typically two types of addition reactions – electrophilic addition and nucleophilic addition. Michael Addition is a type of nucleophilic addition reaction [1-4].
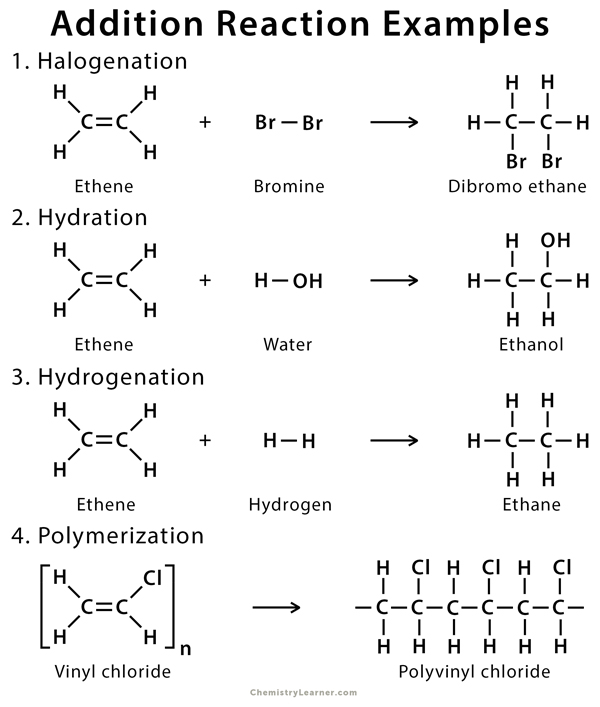
Examples of Addition Reaction
Examples of addition reactions that are common in alkene include halogenation, hydration, hydrohalogenation, hydrogenation, and polymerization [1].
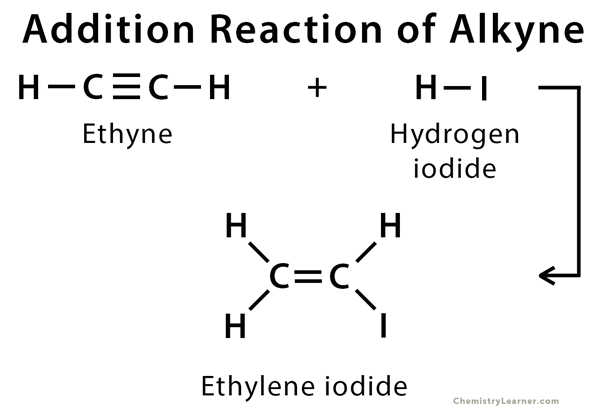
Alkyne can undergo addition reaction to give an alkene or its derivative.
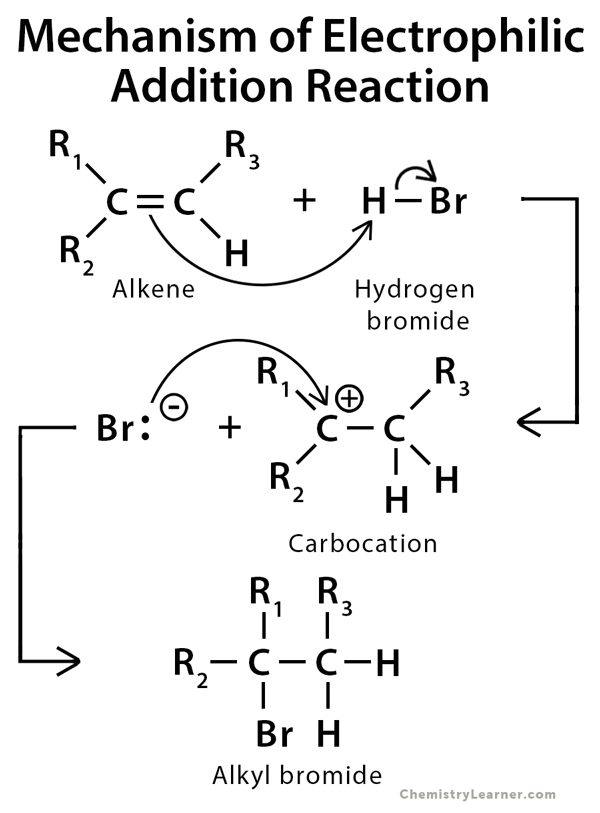
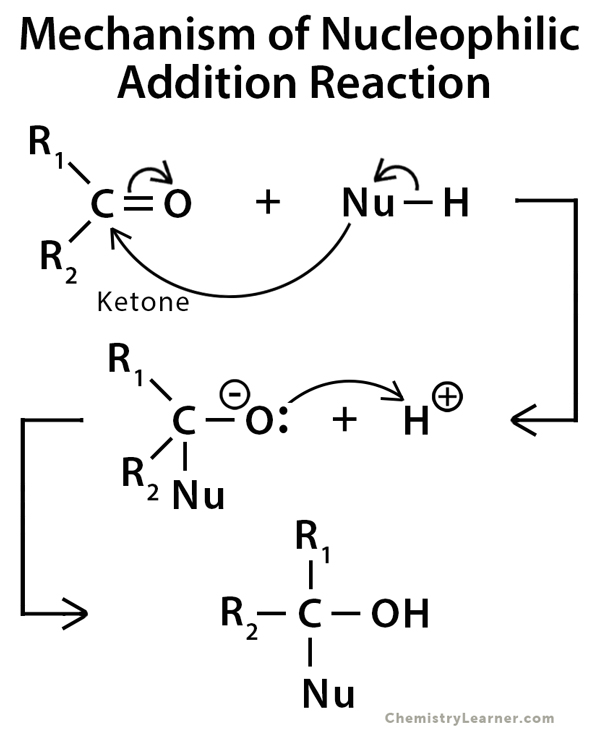
Mechanism of Addition Reaction
The mechanisms of electrophilic and nucleophilic addition reactions proceed through intermediates.
References
- Definition and example – Chem.libretexts.org
- Definition – Chem.ucalgary.ca
- Definition – Chem.ucla.edu
- Definition – Chemistry.msu.edu





Thank you