Alkyne Reactions
Alkynes are highly reactive hydrocarbons that consist of at least one triple bond. The triple bond consists of one sigma (σ) and two pi (π) bonds. The carbon-carbon pi bond is formed by side-by-side overlapping of p orbitals, each from a different atom. The pi bonds are easily broken, and the valence electrons are set free. They make the molecule electron-rich, attract electrophiles, and form new bonds with other atoms. Therefore, alkynes produce various compounds [1-4].
Types of Alkyne Reactions [1-5]
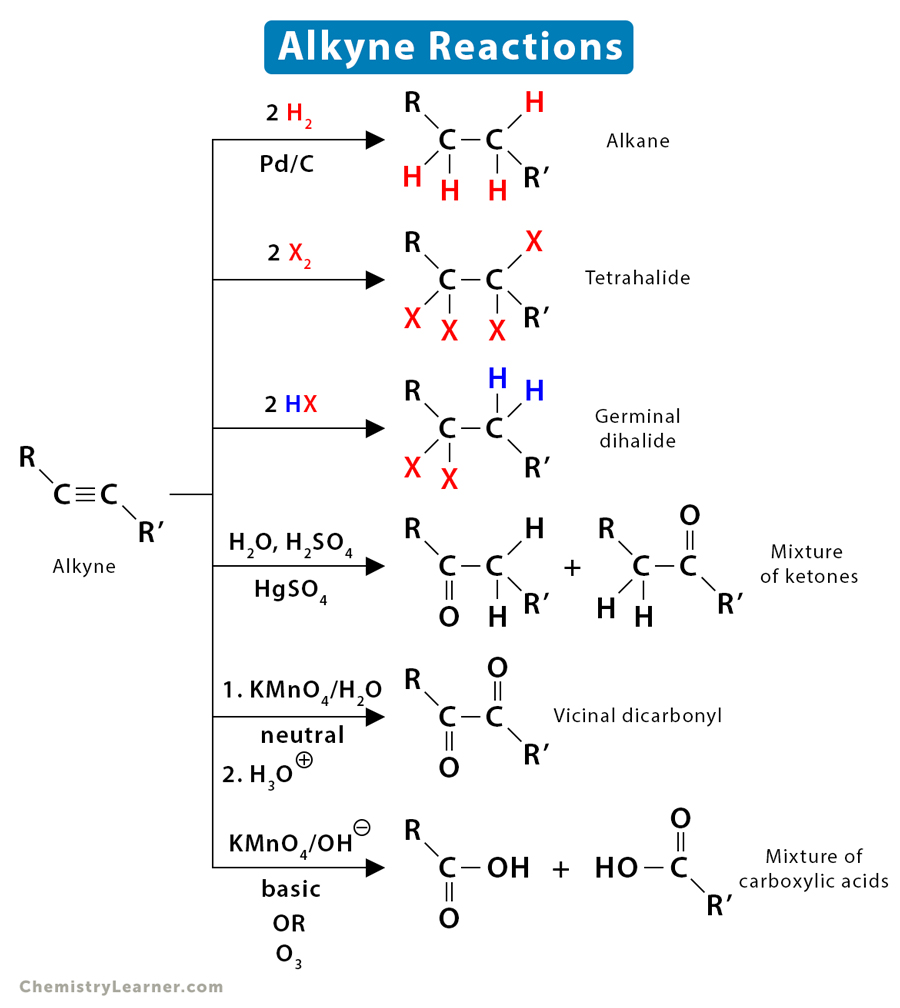
1. Addition
The most common reaction of alkynes is the addition reaction. This reaction involves breaking the pi bond and adding two atoms to the molecule.
General Formula for Addition Reaction of Alkyne
R1 – C ≡ C – R2 + A – B → R1 – AC = CB – R2
Types of Addition Reaction of Alkyne
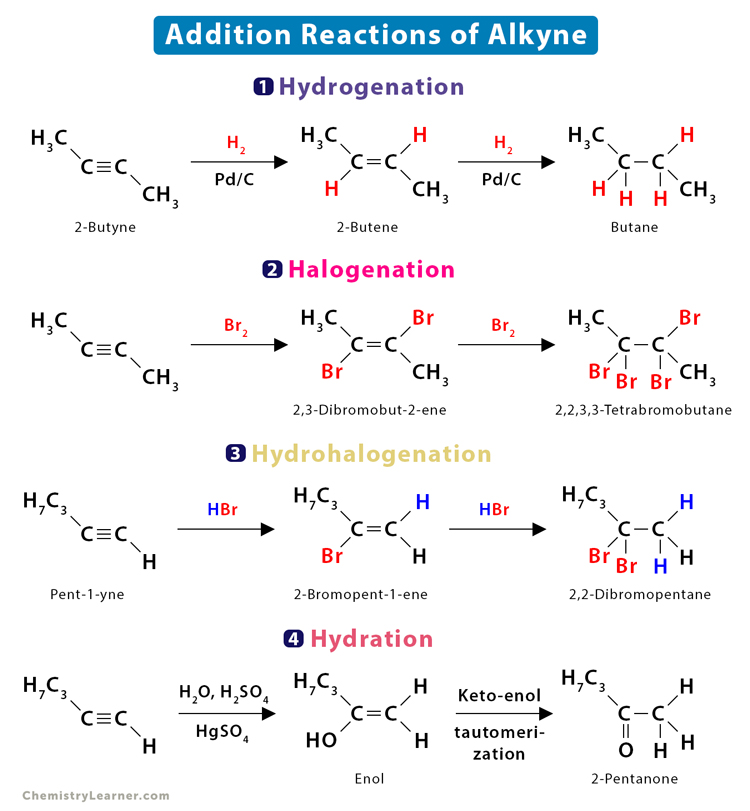
1.1 Hydrogenation
Hydrogenation is a process in which a hydrogen molecule is added to the pi bond. Alkynes undergo hydrogenation in the presence of a catalyst, like platinum or palladium, to form alkenes. Alkenes further undergo hydrogenation to form alkanes. The mechanism of the alkyne hydrogenation is similar to that of alkene. The catalyst absorbs the two hydrogen atoms and adds to the compound in a syn addition.
Examples
- 2-Butyne (C4H6) reacts with hydrogen (H2) in the presence of a Pd/C catalyst to form 2-butene (C4H8), which is then reduced to butane (C4H10).
CH3 – C ≡ C – CH3 + H2 → CH3 – HC = CH – CH3
CH3 – HC = CH – CH3 + H2 → CH3 – H2C – CH2 – CH3
- Phenylacetylene (Ph – C ≡ CH) undergoes hydrogenation in the presence of a Pd/C catalyst to form polystyrene (Ph – HC = CH2), which is further reduced to ethylbenzene (Ph – H2C – CH3).
Ph – C ≡ CH + H2 → Ph – HC = CH2
Ph – HC = CH2 + H2 → Ph – H2C – CH3
1.2 Halogenation
Halogenation is the process of adding a halogen molecule to the pi bond. When halogens are added to alkynes, the result is dihaloalkenes. The alkenes undergo further reaction to give tetrahaloalkane. The mechanism is generally very complex and proceeds through [2+1] cycloaddition.
Examples
- But-2-yne (C4H6) reacts with excess bromine (Br2) to give an intermediate 2,3-dibromobut-2-ene (C4H6Br2), which further reacts with another Br2 molecule to give 2,2,3,3-tetrabromobutane (C4H6Br4).
CH3 – C ≡ C – CH3 + Br2 → CH3 – BrC = CBr – CH3
CH3 – BrC = CBr – CH3 + Br2 → CH3 – Br2C – CBr2 – CH3
- 1-phenyl-1-propyne (Ph – C ≡ C – CH3) reacts with chlorine (Cl2) in presence of CCl4 to give 1,2-dichloro-1-phenyl-1-propene (Ph – ClC = ClC – CH3) which undergoes further chlorination to give 1,1,2,2–tetrachloro-1-phenylpropane (Ph – Cl2C – Cl2C – CH3).
Ph – C ≡ C – CH3 + Cl2 → Ph – ClC = CCl – CH3
Ph – ClC = CCl – CH3 + Cl2 → Ph – Cl2C – CCl2 – CH3
1.3 Hydrohalogenation
Hydrohalogenation refers to adding a hydrogen halide to the pi bond. Adding a hydrogen halide molecule to pi bonds is known as hydrohalogenation. The result is haloalkene, followed by dihaloalkane per Markovnikov’s rule.
Examples
- When hydrogen bromide (HBr) is added to pent-1-yne (C3H7 – C ≡ CH + HBr), the result is 2-bromopent-1-ene (C3H7 – BrC = CH2). This compound reacts with a second molecule of HBr to form 2,2-dibromopentane (C3H7 – Br2C – CH3).
C3H7 – C ≡ CH + HBr → C3H7 – BrC = CH2
C3H7 – BrC = CH2 + HBr → C3H7 – Br2C – CH3
- When hydrogen chloride (HCl) is added to hex-3-yne (C2H5 – C ≡ C – C2H5), the result is 3-chloro-hex-3-ene (C2H5 – ClC = CH – C2H5), which reacts with another molecule of HCl to form 3,3-dichlorohexane (C2H5 – Cl2C – CH2 – C2H5).
C2H5 – C ≡ C – C2H5 + HCl → C2H5 – ClC = CH – C2H5
C2H5 – ClC = CH – C2H5 + HCl → C2H5 – Cl2C – CH2 – C2H5
1.4 Hydration
The addition of a water molecule to the pi bond is known as hydration. This process is carried out in the presence of mercuric sulfate. Unlike alkene, alcohol is not formed because of enol-ketone tautomerization. When water is added, the initial product is an enol, which then rearranges to a more stable keto tautomer.
Examples
- Pent-1-yne (C3H7 – C ≡ CH) is hydrated to give an enol (C3H7 – C(OH) = CH2), which then tautomerizes to 2-pentanone (C3H7 – CO – CH3).
C3H7 – C ≡ CH + H2O → C3H7 – C(OH) = CH2 → C3H7 – CO – CH3
- Acetylene (HC ≡ CH) is hydrated to give acetaldehyde (H3C – CHO).
HC ≡ CH + H2O → H3C – CHO
- Propyne (CH3 – C ≡ CH) is hydrated to give acetone (CH3 – CO – CH3)
CH3 – C ≡ CH → CH3 – CO – CH3
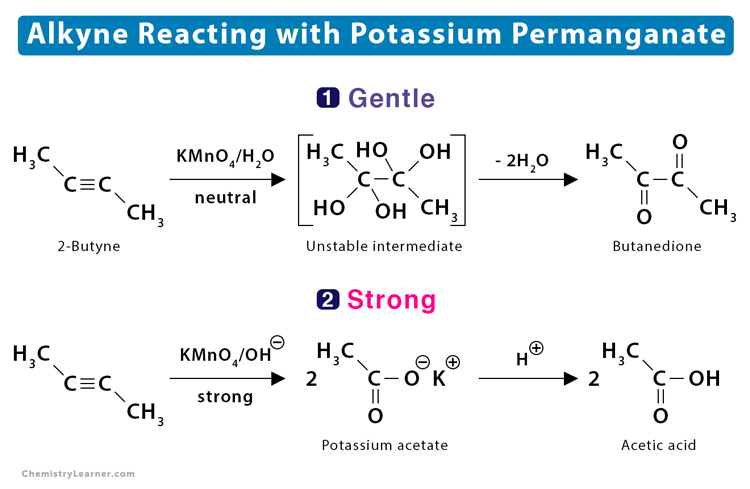
2. Oxidation
Alkynes react with oxidizing agents like potassium permanganate resulting in products depending on the reaction conditions – gentle or strong. For example, alkynes form vicinal dicarbonyl compounds with neutral potassium permanganate (gentle) and carboxylic acid with basic potassium permanganate (strong). The latter is a result of oxidative cleavage.
Example
- The reaction between but-2-yne (CH3 – C ≡ C – CH3) and neutral potassium permanganate (KMnO4) yields 2,3-butadione.
CH3 – C ≡ C – CH3 + KMnO4 → CH3 – OC – CO – CH3
Under more vigorous conditions, this reaction yields acetic acid.
CH3 – C ≡ C – CH3 + KMnO4 → 2 CH3 – COOH
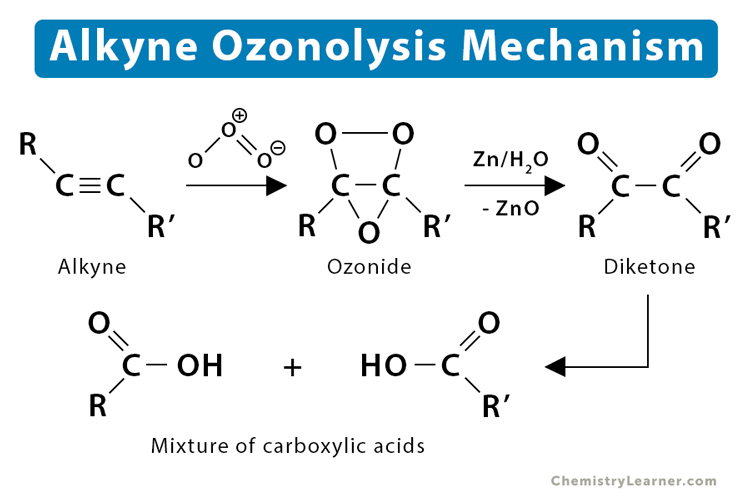
3. Ozonolysis
Ozonolysis is the process by which an ozone molecule is added to an unsaturated compound to cleave its double or triple bond. Ozonolysis of alkyne results in a mixture of carboxylic acids.
Example
- Pent-2-yne reacts with ozone to produce a mixture of acetic and propanoic acids.
C2H5 – C ≡ C – CH3 + O3 → CH3 – COOH + C2H5 – COOH
4. Other Reactions of Alkyne
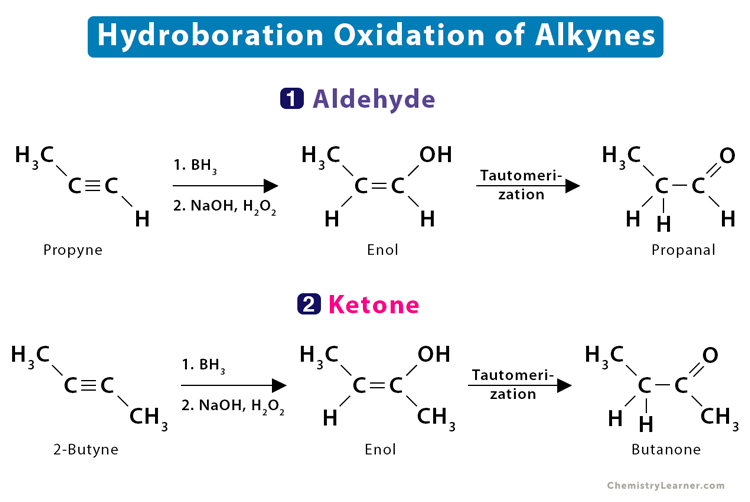
4.1 Hydroboration Oxidation
In this reaction, an alkyne is treated with borane followed by oxidation with alkaline hydrogen peroxide to form an enol. The unstable enol quickly transforms into a more stable aldehyde or ketone.
Example
- Propyne is transformed into propanal
CH3 – C ≡ CH → CH3 – HC = C(OH) – H → CH3 – CH2 – CHO
- But-2-yne is transformed into 2-butanone
CH3 – C ≡ C – CH3 → CH3 – (OH)C = CH – CH3 → CH3 – CO – CH2 – CH3
4.2 Hydroxyhalogenation
Hydroxyhalogenation is halogenation carried out in the presence of excess water. In this case, the nucleophilic hydroxide and the halogen attach to the carbon atoms to give a halohydrin intermediate. The halohydrin is an enol and undergoes keto-enol tautomerization to give halogenated ketone.
Example
- Phenylacetylene (Ph – C ≡ CH) reacts with chlorine (Cl2) in the presence of excess water to give 2-chloroacetophenone (Ph – OC – CH2Cl).
Ph – C ≡ CH + Cl2/H2O → Ph – (OH)C = CHCl → Ph – OC – CH2Cl
4.3 Alkoxyhalogenation
Alkoxyhalogenation is also a halogenation process that is carried out in the presence of excess alcohol. The double addition of the alkoxy and the halogen group gives an acetal.
Example
- Phenylacetylene (Ph – C ≡ CH) reacts with chlorine (Cl2) in the presence of excess ethanol (EtOH) to give (1,1-dichloro-2,2-diethoxy ethyl)benzene.
Ph – C ≡ CH + Cl2/EtOH → Ph – EtC = CHCl → Ph – (EtO)2C – CHCl2
4.4 Deprotonation
The terminal alkynes are highly acidic and can be deprotonated by a strong base like sodium hydride (NaH) or sodium amide (NaNH2). The reaction is nucleophilic substitution.
Example
- Deprotonation of propyne (CH3 – C ≡ CH) gives sodium propynylide (CH3 – C ≡ C– Na+)
CH3 – C ≡ CH + NaNH2 → CH3 – C ≡ C– Na+ + NH3
FAQs
Ans. Only terminal alkynes react with Grignard reagent since they contain acidic hydrogen.