Autoionization (Self-ionization) of Water
The autoionization or self-ionization of water is an endothermic ionization reaction where a molecule of pure water gets dissociated into hydrogen ions (H+) and hydroxyl ions (OH-). As water is a very weak electrolyte, it gets ionized minimally.
Autoionization Chemical Equation
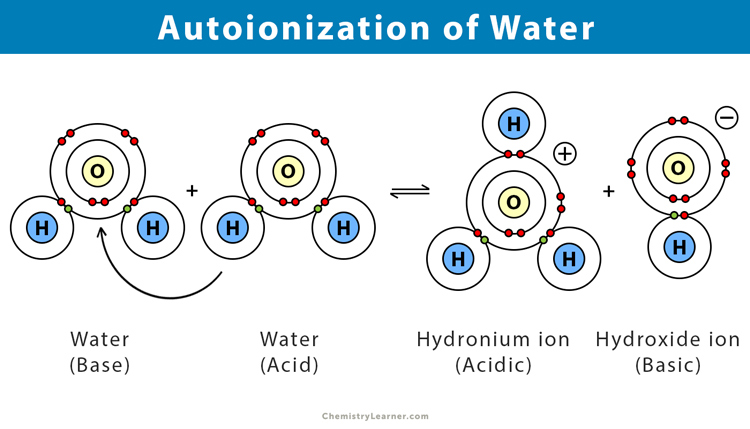
When two water molecules collide, a hydrogen ion from one molecule gets transferred to the other, producing a positively charged hydronium ion (H3O+) and a negatively charged hydroxyl ion.
H2O (l) + H2O (l) ⇌ H3O+ (aq.) + OH– (aq.)
We commonly use the simplified form of the reaction:
H2O (l) ⇌ H+ (aq.) + OH– (aq.)
Acid-Base Properties of Water
As it is amphoteric, water can act as both an acid and a base in solution. When mixed with a base, water donates hydrogen ions and acts as an acid. However, when water is mixed with an acid, it accepts hydrogen ions and acts as a base.
- Acting as a base:
H2O (l) + CH3COOH (aq.) ⇌ H3O+ (aq.) + CH3COO– (aq.)
- Acting as an acid:
NH3 (aq.) + H2O (l) ⇌ NH4+ (aq.) + OH– (aq.)
During autoionization, water can be both an acid and a base to itself.
H2O (l) + H2O (l) ⇌ H3O+ (aq.) + OH– (aq.)
Autoionization Constant
As mentioned before, H2O splits into H+ and OH–.
Kc = [H+] [OH–]/[H2O]
Kc is the dissociation constant of water at equilibrium.
As pure water is a poor electrolyte, it gets slightly ionized. So, the concentration of H2O is much greater than the concentrations of H+ and OH–. Therefore, [H2O] can be regarded as constant.
Kc [H2O] = [H+][OH–]
As Kc and [H2O] are constants, they are replaced by another constant, ‘Kw’.
Kw = [H+][OH–]
Kw is called the autoionization constant of water.
From the above explanation, ‘Kw’ can be regarded as the ionic product of water, i.e., the product of the molar concentrations of H+ and OH– ions. As H+ ions exist as H3O+ ions in water, the ionic product can alternatively be stated as the product of molar concentrations of H3O+ and OH– ions.
Kw = [H3O+][OH–]
Relationship Among pH, pOH, and pKw
The autoionization constant is temperature-dependent and increases with an increase in temperature. It has a fixed value of 1×10-14 at 25 ͦC.
The concentrations of dissociated H+ and OH– ions are equal. So, let the [H+] and [OH–] be ‘x’.
By substituting the values in the equilibrium expression, we get,
[H+] [OH–] = 1 x 10-14
=> x . x = 1 x 10-14
=> x2 = 1 x 10-14
=> x = 1 x 10-7
Therefore, [H+] = [OH–] = 1 x 10-7
Taking negative logarithm on both sides of [H+] equation:
-log [H+] = -log (10-7)
=> pH = – (-7)
=> pH = 7 [ Since, pH = -log [H+]]
Similarly, pOH will also be 7. This result is expected since the pH of pure water is 7.
Now, taking the negative logarithm of Kw
-log (Kw) = -log (1×10-14)
=> pKw = – (-14) = 14
Therefore, at 25 ͦC,
pKw = pH + pOH
References
- Water autoionization and Kw – Khanacademy.org
- Autoionization of Water – Courses.lumenlearning.com
- Self-Ionization of Water and the pH Scale – Chem.libretexts.org