Benzene Ring
Benzene belongs to the family of aromatic compounds with a ring structure. It is the simplest aromatic hydrocarbon consisting of six carbon atoms bonded with alternative single and double bonds. Each of the carbon atoms is singly bonded to a hydrogen atom. The six conjugate carbon atoms and p-orbital delocalization due to sp2 hybridization give rise to a resonance structure [1-4].
Benzene is a colorless liquid with a distinct odor. Its boiling point is 80.1 ˚C. English scientist Michael Faraday discovered it in 1825 from the oily residue left by producing illuminating gas. German organic chemist Friedrich August Kekulé published a paper on its structure in 1865.
Characteristics [1-3]
- A planar molecule with six carbon atoms arranged in a hexagon
- Each carbon atom shares a single bond with a hydrogen atom, giving rise to the chemical formula C6H6
- Carbon atoms alternatively share single and double bonds and hence, have a bond length somewhere between single and double bonds
- Delocalized pi electrons are present above and below the plane of the ring
- Benzene cannot participate in addition reaction since adding compounds would break the ring, and benzene would lose its stability
Structure
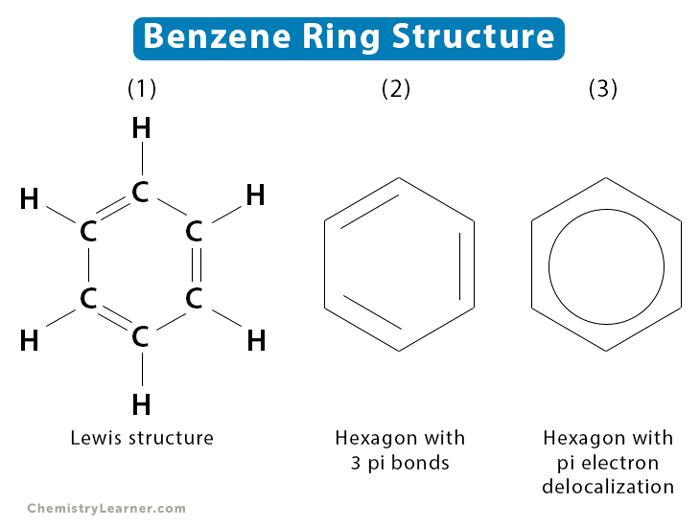
Benzene has a molecular formula, C6H6, whose structure is shown below. There are three ways to illustrate its structure [1-3].
As shown in illustration (1) below, benzene has six carbon atoms arranged in a ring. Single and double bonds alternately separate the carbon atoms. This kind of atomic arrangement is known as a conjugated structure. Each carbon atom is singly bonded with a hydrogen atom. This structure is also known as the Lewis dot structure of benzene.
Another way of representing benzene is the illustration (2). Here, we see a hexagon with each edge representing one carbon atom. The three double bonds are also shown. However, the hydrogen atoms are not shown.
Illustration (3) represents a hexagon with a circle inside. The circle represents the delocalized pi electrons. The structural formula of benzene is often represented by this structure.
Bond Length and Bond Angle
The C-C bond length in benzene is 1.39 Å, and each bond angle is 120 ˚. The bond length value is between the C-C single bond (1.47 Å) and the C≡C triple bond (1.34 Å). The pi electron delocalization makes the bond count as one and a half bond between the carbon atoms. Moreover, experiments have shown that benzene’s bond length is between single and double bond values [1].
FAQs
Ans. There are four degrees of unsaturation in a benzene ring.
Ans. Kekulé pictured benzene’s ring structure after dreaming of a snake eating its tail.