Intermolecular Forces
Table of Contents
What are Intermolecular Forces
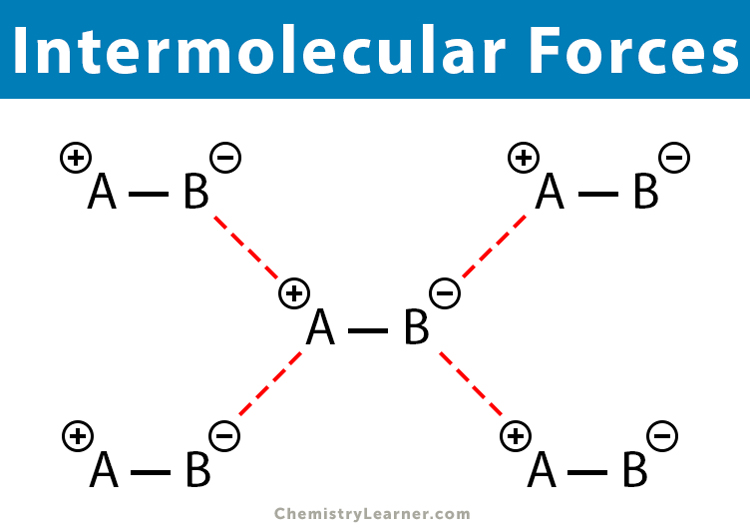
Intermolecular forces, also known as intermolecular interactions, are the electrostatic forces of attraction between molecules in a compound. The intermolecular forces tend to attract the molecules together, bring them closer, and make the compound stable.
Intermolecular forces are important because they affect the compound’s physical properties and characteristics like melting point, boiling point, vapor pressure, viscosity, solubility, and enthalpy. When a substance goes from one state of matter to another, it goes through a phase change. Intermolecular forces play a crucial role in this phase transformation.
How to Determine Intermolecular Forces in Compounds
The polarity of the molecules helps to identify intermolecular forces. A molecule is said to be polar if there is a significant electronegativity difference between the bonding atoms. As a result, one atom will pull the shared electron pairs towards itself, making it partially negative and the other atom partially positive. The distribution of charges in molecules results in a dipole, which leads to strong intermolecular forces.
On the other hand, atoms that do not have any electronegativity difference equally share the electron pairs. The molecules are said to be nonpolar. They interact differently from the polar molecules.
By knowing whether a molecule is polar or nonpolar, one can find the type of intermolecular force.
Types of Intermolecular Forces of Attraction
A compound may have more than one type of intermolecular force, but only one of them will be dominant. All intermolecular forces are known as van der Waals forces, which can be classified as follows.
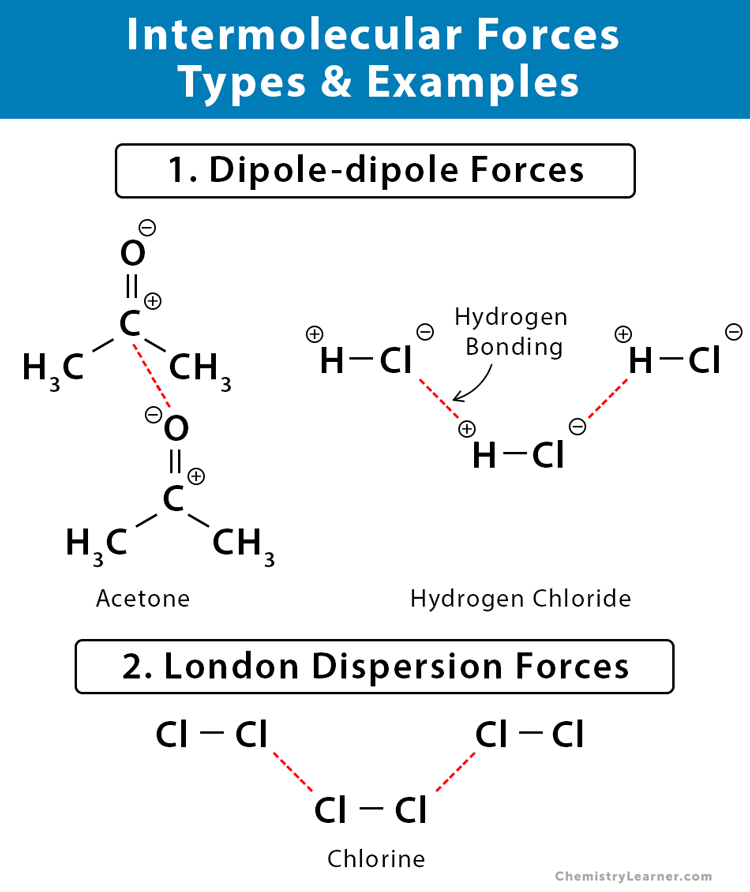
1. Dipole-dipole Forces
They occur between any two molecules that have permanent dipoles. The partially positive end of one molecule is attracted to the partially negative end of another molecule. As a result, the molecules come closer and make the compound stable.
Example: Hydrogen (H2), iodine monochloride (ICl), acetone (CH3)2O, hydrogen sulfide (H2S), difluoromethane (CH2F2), chloroform (CHCl3), hydrogen cyanide (HCN), and phosphine (PH3)
Hydrogen Bonding
It is a particular type of dipole-dipole force. It occurs when a polar molecule consisting of partially positive hydrogen (H) atom is attracted to a partially negative atom of another molecule. This kind of force is seen in molecules where the hydrogen is bonded to an electronegative atom like oxygen (O), nitrogen (N), fluorine (F), chlorine (Cl), bromine (Br), and iodine (I). The hydrogen bond is the strongest intermolecular force.
Examples: Water (H2O), hydrogen chloride (HCl), ammonia (NH3), methanol (CH3OH), ethanol (C2H5OH), and hydrogen bromide (HBr)
2. London Dispersion Forces
They occur in nonpolar molecules held together by weak electrostatic forces arising from the motion of electrons. When electrons move around a neutral molecule, they cluster at one end resulting in a dispersion of charges. As a result, a temporary dipole is created that results in weak and feeble interactions with other molecules. This type of force is observed in condensed phases like solid and liquid. London dispersion force is the weakest intermolecular force.
Examples: Chlorine (Cl2), oxygen (O2), nitrogen (N2), carbon dioxide (CO2), methane (CH4), carbon tetrachloride (CCl4), hexane (C6H6), silane (SiH4), hydrogen cyanide (HCN), phosphine (PH3), carbon disulfide (CS2), and ethane (CH3CH3)
Strength of Intermolecular Forces
Substances with high intermolecular forces have high melting and boiling points. The reason is that more energy is required to break the bond and free the molecules. Liquids with high intermolecular forces have higher surface tensions and viscosities than liquids with low ones. The stronger the intermolecular forces between solute and solvent molecules, the greater the solubility of the solute in the solvent. Elastomers have weak intermolecular forces. Volatile substances have low intermolecular force.
Ranking
The strength of intermolecular force from strongest to weakest follows this order:
Hydrogen bonding > Dipole-dipole forces > London dispersion forces
The following table compares the different intermolecular forces and shows their effects on the melting and boiling points of substances.
| Intermolecular force | Melting point | Boiling point | |
|---|---|---|---|
| Methane (CH4) | London dispersion | -182 ͦC | -164 ͦC |
| Acetone (CH3)2O | Dipole-dipole | -95 ͦC | 56 ͦC |
| Water (H2O) | Hydrogen bonding | 0 ͦC | 100 ͦC |
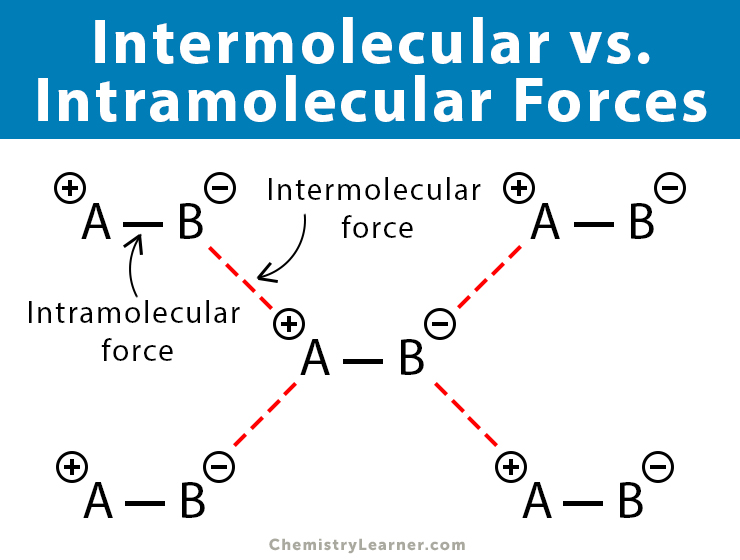
Intermolecular Forces vs. Intramolecular Forces
While intermolecular forces take place between the molecules, intramolecular forces are forces within a molecule. Chemical bonds are intramolecular forces between two atoms or two ions. Oppositely charged ions attract each other and complete the (ionic) bond. On the other hand, atoms share electrons with other atoms to complete the (covalent) bond.
The intermolecular forces are entirely different from chemical bonds. Unlike bonds, they are weak forces. The table below compares and contrasts inter and intramolecular forces.
| Intermolecular forces | Intramolecular forces | |
|---|---|---|
| How are they formed | Between molecules in a compound | Within the atoms or ions of a molecule |
| Strength | Weak (1 – 12 kJ/mole) | Strong (50 – 200 kJ/mole) |
| Effect on substance | Physical properties like melting point, boiling point, and solubility | Chemical properties |
| Nature | Attractive or repulsive | Chemical bonds (Intramolecular hydrogen bond is also possible) |
| Types | Dipole-dipole forces, hydrogen bonding, and London dispersion forces | Ionic bonds, covalent bonds, and metallic bonds |
| Examples | Water (H2O), hydrogen chloride (HCl), and chlorine (Cl2) | Sodium chloride (NaCl), potassium iodide (KI), and magnesium oxide (MgO) |
FAQs
Ans. The dispersion force is present in all atoms and molecules, whether they are polar or not.
Ans. Hydrogen bonding is the dominant intermolecular force in water (H2O).
Ans. Water has a stronger intermolecular force than isopropyl alcohol since it takes longer to evaporate.
References
- Intermolecular forces – Chem.libretexts.org
- Intermolecular Bonding – van der Waals Forces – Chemguide.co.uk
- Intermolecular Forces – Chemed.chem.purdue.edu
- Intermolecular Forces – Opentextbc.ca
- Intermolecular Forces – Chem.ucalgary.ca
- Intermolecular Forces – Chemistry.msu.edu