Dihydroxylation
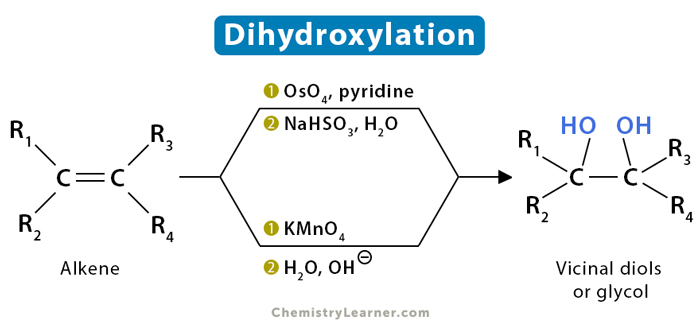
Dihydroxylation is an oxidation process when an alkene is converted into a vicinal diol, also known as 1,2-diols or glycols. The most frequent and direct process of dihydroxylation is using a transition metal in a high oxidation state, typically osmium (VIII) or manganese (VII). The metal is often used as a catalyst in a compound, like osmium tetraoxide (OsO4) or potassium permanganate (KMnO4) [1-4].
There are two different stereochemical methods of dihydroxylation – syn and anti. In a syn-addition, the two substituents are added to the same side (or face) of a double bond. In anti-addition, the two substituents are added to the opposite side.
Example [1-4]
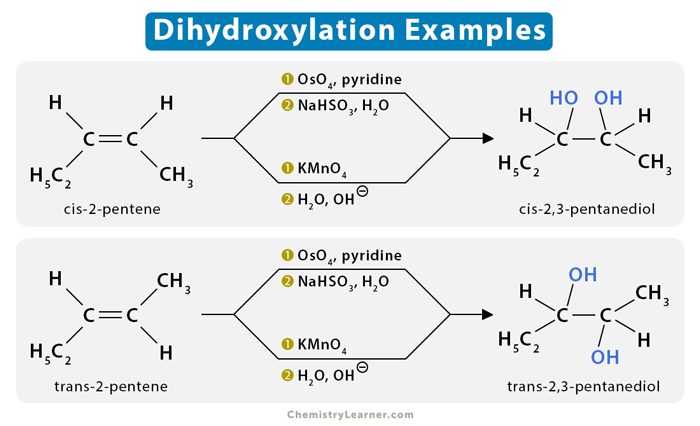
Syn-dihydroxylation
cis-2-pentene (C2H5CH=CHCH3) reacts with osmium tetroxide (OsO4) in the presence of potassium bisulfate (NaHSO3) to give cis-2,3-pentanediol. Likewise, trans-2-pentene gives trans-2,3-pentanediol.
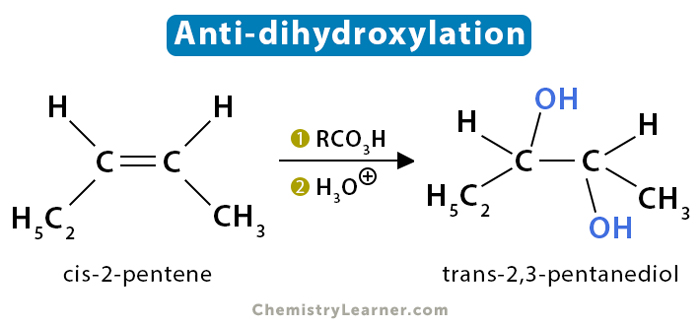
Anti-dihydroxylation
cis-2-pentene (C2H5CH=CHCH3) reacts with a peroxyacid such as meta-chloroperoxybenzoic acid (mCPBA) to give trans-2,3-pentanediol.
Mechanism
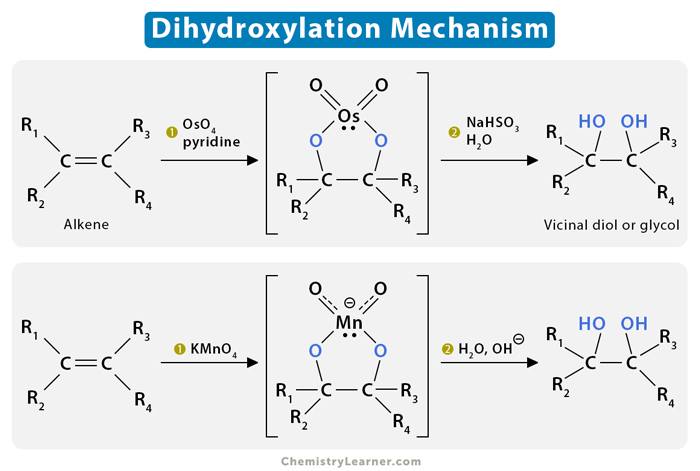
The mechanism of alkene dihydroxylation is a two-step concerted cycloaddition reaction. We shall look at the syn- and anti-dihydroxylation mechanisms separately.
Syn-dihydroxylation
Osmium tetroxide is an electrophilic compound and a powerful oxidizing agent. It accepts an electron pair from the alkene π bond while the osmium reduces from +VIII to +VI.
Step 1: The C-C pi bond combines with two Os=O bonds to give a five-membered ring structure known as an osmate ester.
Step 2: The intermediate cyclic ester is isolated and reacts with a reducing agent like sodium bisulfite that cleaves the Os–O bond and produces a cis-glycol while retaining the syn stereochemistry of the two newly formed C–O bonds.
Osmium tetroxide is very expensive and is often used as a catalyst along with other oxidizing agents like N-methylmorpholine N-oxide (NMO) or tert-butyl hydroperoxide (TBHP). The co-oxidant reoxidizes the osmium +6 to +8, thereby regenerating osmium tetroxide for further oxidation of the remaining alkenes.
Anti-dihydroxylation
Step 1
The anti-dihydroxylation of alkene is achieved by converting the alkene into an epoxide first using a peroxyacid such as meta-chloroperoxybenzoic acid (mCPBA). The oxygen in the peroxide is electron-deficient and is attacked by the electrons of the π bond. The O-O bond is relatively weak, which also makes the bond cleavage easier.
Step 2
Epoxides are highly strained three-membered rings that are very reactive. The epoxide ring can be opened under acidic conditions. The acid catalyzes by converting the oxygen into a good leaving group. The result is a product consisting of two hydroxyl groups attached to two adjacent carbon atoms – a trans-diol
The mechanism of epoxide hydrolysis is discussed in our article on epoxide reactions.