Formal Charge of Ozone (O3)
The ozone (O3) molecule consists of three oxygen (O) atoms. The central oxygen (O) atom forms a single covalent bond with another oxygen atom and a double covalent bond with the third. It has two resonance structures that are shown below.
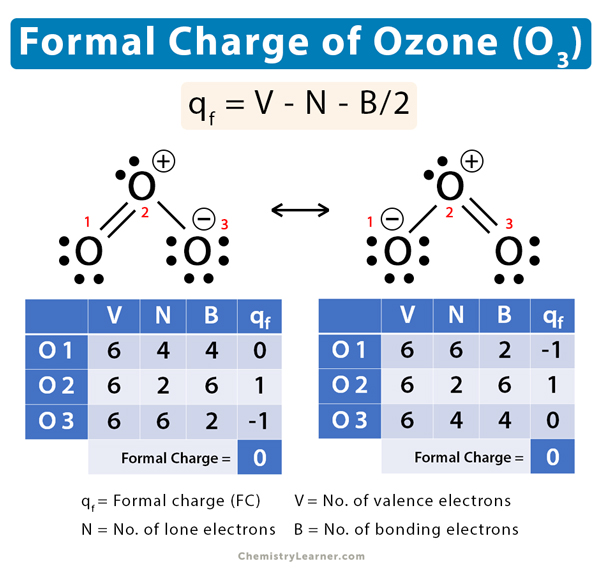
O=O+-O– ↔ O–-O+=O
To determine the formal charge of ozone, we must first calculate the formal charges on the three oxygen atoms.
i. O=O+-O–
Oxygen 1
V = 6, N = 4, B = 4
Therefore,
qf = 6 – 4 – 4/2 = 0
Oxygen 2
V = 6, N = 2, B = 6
Therefore,
qf = 6 – 2 – 6/2 = 1
Oxygen 3
V = 6, N = 6, B = 2
Therefore,
qf = 6 – 6 – 2/2 = -1
The net formal charge is: 0 + 1 – 1 = 0
ii. O–-O+=O
Oxygen 1
V = 6, N = 6, B = 2
Therefore,
qf = 6 – 6 – 2/2 = -1
Oxygen 2
V = 6, N = 2, B = 6
Therefore,
qf = 6 – 2 – 6/2 = 1
Oxygen 3
V = 6, N = 4, B = 4
Therefore,
qf = 6 – 4 – 4/2 = 0
The net formal charge is: -1 + 1 + 0 = 0
The formal charge of both the resonance structures of ozone is zero. It means that both these structures are stable and contribute equally to the molecule’s overall hybrid structure.