Oxygen
What is Oxygen
Oxygen (pronunciation: OK-si-jen) is a colorless element that belongs to the group of Chalcogens in the periodic table, and it is represented by the chemical symbol O [1, 2, 3]. A highly reactive non-metal, it can easily form oxides with most of the other elements and their compounds [3]. At room temperature, it is an odorless, tasteless diatomic gas characterized by the formula O2 [3].
Isotopes
Oxygen has three-known stable isotopes, including 16O, 17O, and 18O with a natural abundance of 99.76%, 0.04%, and 0.20% respectively [4]. Radioactive isotopes from 12O to 24O also exist out of which the longest-lived is 15O characterized by a half-life period of 122.24 seconds whereas the shortest-lived 12O has a half-life period of 580(30) X 10-24 seconds [4].
Where is Oxygen Found
It is found in the Earth’s atmosphere, constituting 21% of the atmospheric volume [1]. Oxygen along with its compounds accounts for nearly two-thirds of the mass of the human body and 49.2% of the mass of the Earth’s crust [1].
Oxygen gas is commercially extracted by fractional distillation of liquefied air [1]. It can also be obtained by passing dry air through zeolite, a microporous mineral that can absorb nitrogen but leave oxygen [1]. Passing the air through a selectively permeable ceramic membrane can produce highly pure oxygen [1].
It can artificially be produced in the laboratory by using manganese (IV) oxide as a catalyst to decompose aqueous hydrogen peroxide or by electrolytic decomposition of water [1]. Oxygen is also formed when potassium chlorate (KClO3) is heated strongly [2].
History
Origin of its Name: It is derived from ‘oxy genes’, Greek for “acid forming” [1, 2]
Who discovered it: The English chemist Joseph Priestly and the German-Swedish chemist Carl Wilhelm Scheele are known for their independent discovery of oxygen [1, 2].
When and How was it Discovered
Although several chemists had produced oxygen before its discovery, they all failed to identify it and describe its properties [2]. In 1608, the Dutch engineer Cornelius Drebbel showed that heating potassium nitrate (saltpeter) released a gas, which we now know was oxygen [1].
The credit for the discovery of oxygen can be given to three chemists – Joseph Priestley, Carl Wilhelm Scheele, and Antoine Lavoisier [1]. Having produced oxygen in 1774 in Wiltshire, England by heating mercuric oxide and collecting the gas that was released, Priestley published an account of his discovery [1]. He observed that the gas made breathing easier and caused a candle to burn more brightly [1].
Although Carl Wilhelm Scheele had successfully obtained oxygen in 1771 in Uppsala, Sweden, his written account got published in 1777 much later than that of Priestley [1]. The French chemist Antoine Lavoisier is known for proposing the name oxy-gene because he incorrectly believed that the new gas was essential to the formation of all acids [2].
Oxygen Identification |
|||
| Atomic number | 8 [1] | ||
| CAS number | 7782-44-7 [1] | ||
| Position in the periodic table | Group | Period | Block |
| 16 [1] | 2 [1] | p [1] | |
Properties and Characteristics of Oxygen
General Properties |
||||||||||||||||
| Relative atomic mass | 15.999 [1] | |||||||||||||||
| Atomic mass | 15.999 amu [1] | |||||||||||||||
| Molar mass | 15.99940 ± 0.00030 g/mol [5] | |||||||||||||||
| Molecular weight | 31.998 g/mol [6] | |||||||||||||||
| Mass number | 16 [7] | |||||||||||||||
| Allotropes | O2, O3 [1] | |||||||||||||||
Physical Properties |
||||||||||||||||
| Color | Gas: colorless, Liquid: pale blue [1, 8] | |||||||||||||||
| Melting point/freezing point | -218.79 °C, -361.82 °F [1] | |||||||||||||||
| Boiling point | -182.962 °C, -297.332 °F [1] | |||||||||||||||
| Density | 0.001308 g cm-3 [1] | |||||||||||||||
| State of matter at room temperature (solid/liquid/gas) | Gas [1, 7] | |||||||||||||||
| Thermal (heat) conductivity | 0.02658 W/(m K) [9] | |||||||||||||||
| Specific heat | 918 J kg-1 K-1 [1] | |||||||||||||||
| Bulk modulus | Unknown [1] | |||||||||||||||
| Shear modulus | Unknown [1] | |||||||||||||||
| Young’s modulus | Unknown [1] | |||||||||||||||
| Vapor pressure | ||||||||||||||||
| – Temperature (K) | 400 | 600 | 800 | 1000 | 1200 | 1400 | 1600 | 1800 | 2000 | 2200 | 2400 | |||||
| – Pressure (Pa) | – | – | – | – | – | – | – | – | – | – | – | |||||
Chemical Properties |
||||||||||||||||
| Oxidation state/Oxidation number | -2, -1, +1, +2 [1] | |||||||||||||||
| Isotopes | Isotope | Mass | Abundance (%) | Half-life | Mode of decay | |||||||||||
| 16O | 15.995 | 99.757 | – | – | ||||||||||||
| 17O | 16.999 | 0.038 | – | – | ||||||||||||
| 18O | 17.999 | 0.205 | – | – | ||||||||||||
Atomic Data of Oxygen (Element 8)
| Valence electrons | 6 [10] | |||||||
| Quantum numbers | ||||||||
| – n | 2 [11] | |||||||
| – ℓ | 1 [11] | |||||||
| – mℓ | -1 [11] | |||||||
| – ms | -1/2 [11] | |||||||
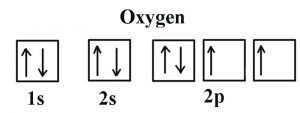
| Electron configuration (noble gas configuration) | [He] 2s22p4 [1] | |||||||
| Atomic structure | ||||||||
| – Number of electrons | 8 [7] | |||||||
| – Number of neutrons | 8 [7] | |||||||
| – Number of protons | 8 [7] | |||||||
| Radius of Atom | ||||||||
| – Atomic radius | 1.52 Å [1] | |||||||
| – Covalent radius | 0.64 Å [1] | |||||||
| Electronegativity (Pauling-scale) | 3.44 [1] | |||||||
| Electron affinity | 140.976 kJ mol-1 [1] | |||||||
| Ionization energy (kJ mol-1) | 1st | 2nd | 3rd | 4th | 5th | 6th | 7th | 8th |
| 1313.942 | 3388.671 | 5300.47 | 7469.271 | 10989.584 | 13326.526 | 71330.65 | 84078.3 | |
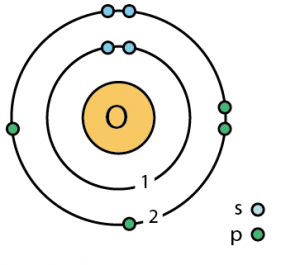
Oxygen Electron Configuration (Bohr Model)
Its Uses
- Oxygen gas is commercially used in the steel industry for removing undesirable compounds and impurities during the forging process [1, 12].
- Large amounts of oxygen are used to manufacture various chemicals like hydrogen peroxide and nitric acid [1]. Epoxyethane, a chemical used for producing antifreeze (chemicals added to water in automobile engines) is manufactured from oxygen [1].
- It can be used for making polyester and chloroethane, which is a precursor to PVC [1].
- The oxy-acetylene flame used in welding or cutting metals contains oxygen gas [1].
- It is now increasingly used for treating sewage and industrial effluent [1].
- Compressed oxygen gas is used in pneumatic tools, including riveters, drills, and paint sprayers [12].
- It is used in pediatric incubators in hospitals to help provide newborn babies the environmental safety they need when they are born prematurely [12].
- Rocket fuel has high oxygen content for increasing the burning rate [12].
- Astronauts, submariners, and scuba divers rely on artificially-delivered oxygen [3]. For deep-sea diving, oxygen is diluted with other gases like helium or nitrogen for preventing oxygen toxicity [3].
Biological Role of Oxygen
The element first appeared 2 billion years ago when photosynthesis of cyanobacteria resulted in a buildup of oxygen in the Earth’s atmosphere [1]
Photosynthesis uses solar energy to divide water molecules into hydrogen and oxygen, from which the oxygen accumulates into the atmosphere while hydrogen along with carbon dioxide produces biomass [1].
Life on Earth now depends on oxygen because all living things need it for respiration and producing energy [1]. Since gaseous oxygen is soluble in water, aerobic bacteria can survive in lakes, rivers, and oceans [1].
Interesting Facts
- Oxygen liquefies at -183.0 °C and can cause frostbite after skin contact [8].
- Its graphical representation indicates its importance and existence in the air, and in combination with hydrogen, in water [1].
- Too little oxygen (below 17% by volume in the Earth’s atmosphere) can make breathing difficult for people while too much (above 25% in the atmosphere) can cause organic compounds to become highly flammable [1].
- Oxygen is produced at the center of stars, with nuclear fusion occurring between carbon-12 and helium-4 [8].
Cost of Oxygen
Canned oxygen usually costs less than $50 per unit [13]. The price of 100 g of pure oxygen is about $0.3 while its price in bulk is $0.02 [14].
- References
- http://www.rsc.org/periodic-table/element/8/oxygen
- https://education.jlab.org/itselemental/ele008.html
- https://courses.lumenlearning.com/boundless-chemistry/chapter/oxygen/
- https://education.jlab.org/itselemental/iso008.html
- https://www.webqc.org/molecular-weight-of-O.html
- https://pubchem.ncbi.nlm.nih.gov/compound/oxygen#section=Top
- https://hobart.k12.in.us/ksms/PeriodicTable/oxygen.htm
- https://www.livescience.com/28738-oxygen.html
- http://periodictable.com/Elements/008/data.html
- https://socratic.org/questions/how-many-valence-electrons-does-oxygen-have
- http://chemistry-reference.com/q_elements.asp?language=en&Symbol=O
- https://sciencing.com/10-uses-oxygen-8634456.html
- Provider.inogen.com
- https://www.chemicool.com/elements/oxygen.html