Gibbs Free Energy
The Gibbs free energy is the energy associated with a chemical process that can be used to do work. It is a state function that is defined in terms of three other state functions – enthalpy, temperature, and entropy. It is denoted by the symbol G. The formula for Gibbs free energy of a system is given by the enthalpy minus the product of the temperature and entropy [1-4].
G = H – TS
Where,
G: Gibbs free energy
H: Enthalpy
T: Temperature
S: Entropy
Unit: kJ/mol or kilo Joule per mole
American chemist J.W. Gibbs developed the free energy concept in the 1870s.
How to Calculate Gibbs Free Energy [1-5]
Change in Gibbs Free Energy
The change in Gibbs free energy is a useful term to determine the spontaneity of a thermodynamic process. It is defined as the difference between the Gibbs free energies of the final and the initial states of the system [2].
ΔG = Gfinal – Ginitial
Consider a chemical reaction where reactants A and B combine to form products C and D. The equation is given by
A + B ⇌ C + D
Reactants ⇌ Products
Then, the change in Gibbs free energy is
ΔG = GProducts – GReactants
This change ΔG depends only on the initial and final values of the free energy and is independent of the path taken. It means that ΔG is independent of the reaction mechanism. From the equation for G, we get
ΔG = ΔH – Δ(TS)
This change is meaningful under two conditions – constant temperature and constant pressure. And, these conditions are typical in a laboratory. Therefore,
ΔG = ΔH – TΔS
Thus, the change in Gibbs free energy is equal to the change in enthalpy minus the product of temperature and change in entropy.
Determination of Spontaneity
The significance of the above equation is to determine the relative importance of the enthalpy and entropy terms as forces that drive a particular chemical reaction. This equation is used to determine the spontaneity of the reaction and predict in which direction it proceeds – forward or backward. Let us look at a few cases where the value of ΔG depends entirely on the values of ΔH and TΔS.
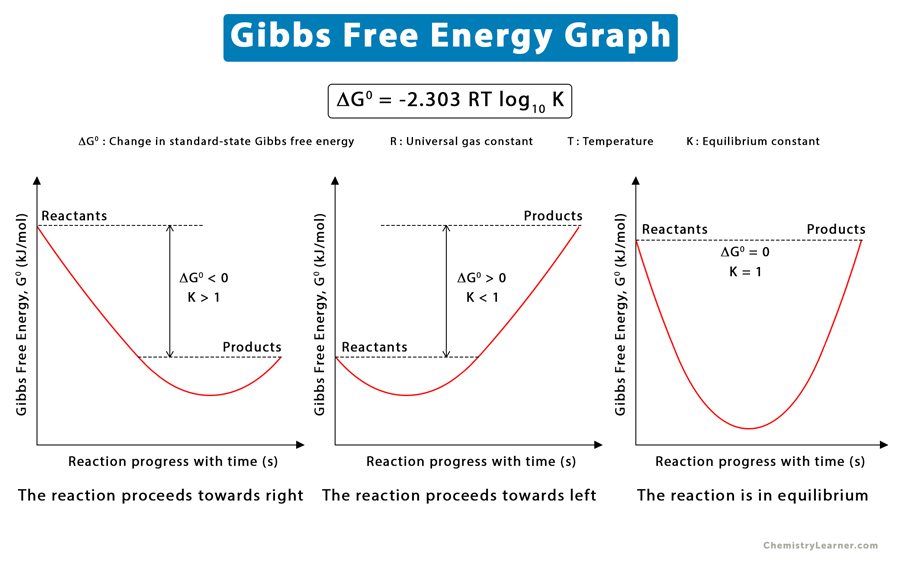
- If ΔG < 0, the reaction is spontaneous and proceeds in the right or forward direction. It is known as spontaneous reaction or exergonic reaction.
- If ΔG > 0, the reaction is nonspontaneous in the forward direction but spontaneous in the reverse direction. Hence, it will proceed toward the left. A nonspontaneous reaction is known as endergonic reaction.
- ΔG = 0, the reaction is in equilibrium.
Standard-State Gibbs Free Energy
As mentioned before, the equation for ΔG applies to constant temperature and pressure conditions. When the reaction takes place at standard temperature and pressure (STP), the equation is rewritten as,
ΔG0 = ΔH0 – TΔS0
G0 is known as the standard-state Gibbs free energy, and ΔG0 is its change. This change can be calculated from the standard-state enthalpy and entropy tables.
National Institute of Standards and Technology (NIST) in Maryland, USA uses a temperature of 20 °C (293.15 K) and an absolute pressure of 1 atm (101.325 kPa) as the standard temperature and pressure. The difference between the change of standard-state Gibbs free energy ΔG0 and the change of Gibbs free energy ΔG is that the reactants and the products are at STP in the former.
Standard Gibbs Free Energy of Formation
The standard Gibbs free energy of formation of a substance is defined as the free energy change when 1 mole of the substance is prepared from its elements at standard temperature and pressure. It is denoted by ΔGf0. The following table shows the standard Gibbs free energy of formation of a few substances.
| Substance | Standard Gibbs Free energy of formation, ΔGf0 (kJ/mol.) |
| Water (liquid) | -237.14 |
| Water (gas) | -228.61 |
| Sodium chloride (solid) | -384.1 |
| Ammonia (gas) | -16.4 |
Gibbs Free Energy at Equilibrium
Let us revisit the following chemical reaction.
A + B ⇌ C + D
As the reaction progresses, the Gibbs free energy changes with time. The change in Gibbs free energy can be expressed in terms of the change in standard-state Gibbs free energy.
ΔG = ΔG0 + RT ln Q
Where,
ΔG: Change in Gibbs free energy
ΔG0: Change in standard-state Gibbs free energy
R: Ideal gas constant (= 8.314 J/mol)
T: Temperature (in K)
Q: Reaction quotient (= [C][D]/[A][B])
Let us look at two different situations.
Case 1:
When Q = 1, ΔG = ΔG0
This point describes the standard-state conditions of the reaction, where ΔG is equal to the change in standard-state Gibbs free energy.
Case 2:
When ΔG = 0, Q = K
There is no driving force for the reaction, and the system is at equilibrium. Here, K is known as the equilibrium constant. Therefore,
0 = ΔG0 + RT ln K
Or, ΔG0 = – RT ln K
ΔG0 = -2.303 RT log10 K
This equation gives the relationship between the standard-state Gibbs free energy and the equilibrium constant. It is used to determine the value of K from ΔG0 and vice versa.
Example of Gibbs Free Energy
Formation of Ammonia (NH3)
Let us take the example of ammonia synthesis to understand Gibbs free energy further [3].
N2 (g) + 3 H2 (g) ⇌ 2 NH3 (g)
The standard Gibbs free energy of formation of two moles of ammonia (NH3) from nitrogen (N2) and hydrogen (H2) is ΔGf0 = -32.8 kJ (see table above). This parameter is calculated when the reaction occurs at standard temperature and pressure. It means that the above reaction takes place at 293.15 K and 1 atm pressure.
The fact that ΔGf0 is negative indicates that the reaction favors the formation of ammonia. In other words, under standard conditions, the reaction proceeds towards the right. The reactants are converted into products before reaching equilibrium. The magnitude of ΔGf0 tells how far the standard-state is from equilibrium. The larger the value, the further the reaction from equilibrium.
Problems and Solutions
Problem 1. Calculate the ΔG for ice melting at 30 ˚C if the enthalpy and entropy of fusion of water are 6 kJ/mol and 22 J/mol-K.
Solution:
The reaction is,
H2O (s) ⇌ H2O (l)
Given,
ΔHfus = 6 kJ/mol = 6000 J/mol
ΔSfus = 22 J/mol-K
T = 30 ˚C = (273 + 30) K = 303 K
Gibbs free energy equation is given by
ΔGfus = ΔHfus – T ΔSfus
Or, ΔGfus = 6000 J/mol – 303 K x 22 J/mol-K
Or, ΔGfus = -666 J/mol
The fact that ΔGfus is negative indicates that the reaction is spontaneous in the forward direction. In other words, ice will melt into the water at 30 ˚C.
Problem 2. ∆G0 for the reaction below is 31.4 kJ/mol at 298.15 K. Use the thermodynamics parameters to decide in which direction the reaction is spontaneous when the concentrations of H2SO4 (aq.), HSO4− (aq.), and H3O+ (aq.) are 0.5 M, 0.25 M, and 0.25 M respectively.
H2SO4 (aq.) + H2O (aq.) ⇌ H3O+ (aq.) + HSO4− (aq.)
Solution:
The reaction quotient is given by,
Q = [H3O+ (aq.)][ HSO4− (aq.)]/[H2SO4 (aq.)]
= 0.25 M x 0.25 M/0.5 M
= 0.125
Given
ΔG0 = 31.4 kJ/mol = 31400 J/mol
T = 298.15 K
R = 8.314 J/mol-K
The change in Gibbs free energy is
ΔG = ΔG0 + RT ln Q
Or, ΔG = 31400 J/mol + 8.314 J/mol-K x 298.15 K x ln (0.125)
Or, ΔG = 26245 J/mol or 26.2 kJ.mol
Since ΔG > 0, the reaction is spontaneous in the reverse direction.