Haworth Projection
Definition: What is Haworth Projection?
Haworth projection is a common way of writing a structural formula of sugars (monosaccharides and disaccharides) with an unpretentious three-dimensional perspective. This type of illustration is mostly used in organic chemistry, especially biochemistry. Haworth projection differs from Fischer projection in that it represents the carbohydrate in its cyclical form. It retains the essential aspects of the molecule, such as the orientation of the hydrogen (H) and hydroxyl (OH) attachments.
Monosaccharides are a 4-, 5- or 6-membered ring structures and have several stereocenters. In order to distinguish between types of monosaccharides (e.g., glucose and fructose), it is essential to have a simple way to draw the sugar molecules while keeping the orientation of the hydroxyl groups. Haworth projection has become the standard in organic chemistry for stereochemical carbohydrate illustrations.
The Haworth projection has been named after the British chemist and Nobel laureate Norman Haworth. His 1929 publication of The Constitution of Sugar has become a classic.
Types of Sugar
Sugars can be classified into monosaccharides and disaccharides. Disaccharides are composed of two monosaccharide units.
Monosaccharide
Four Carbon Aldehyde or Aldotetrose: Throse and erythrose.
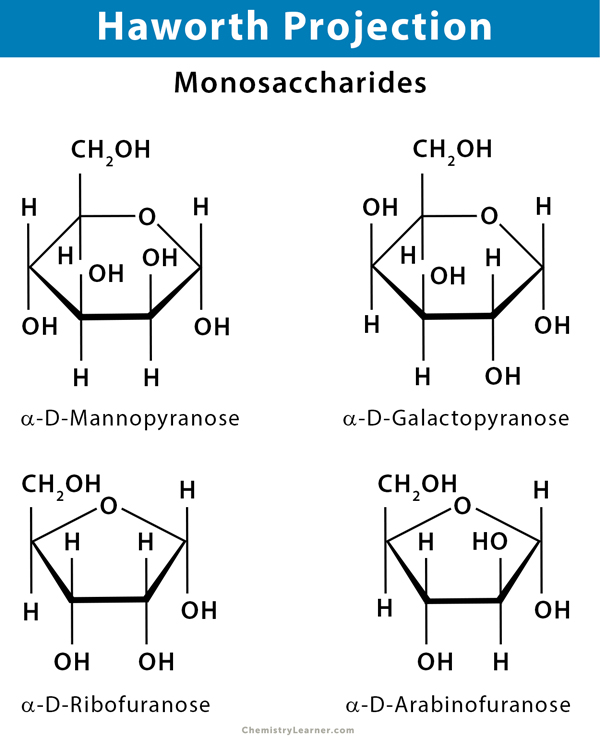
Five Carbon Aldehyde or Aldopentose: Ribose, arabinose, xylose, and lyxose
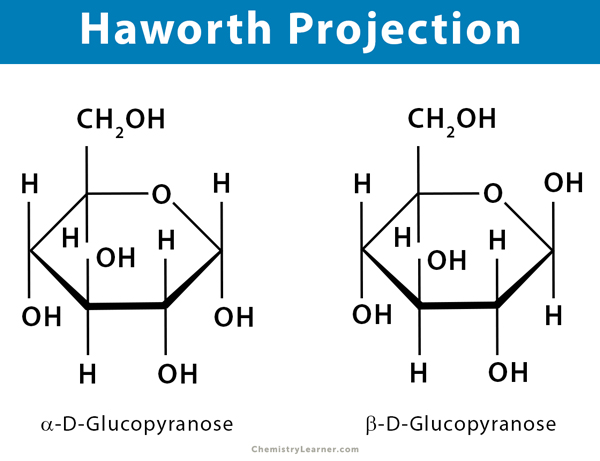
Six Carbon Aldehyde or Aldohexose: Glucose, galactose, mannose, and allose
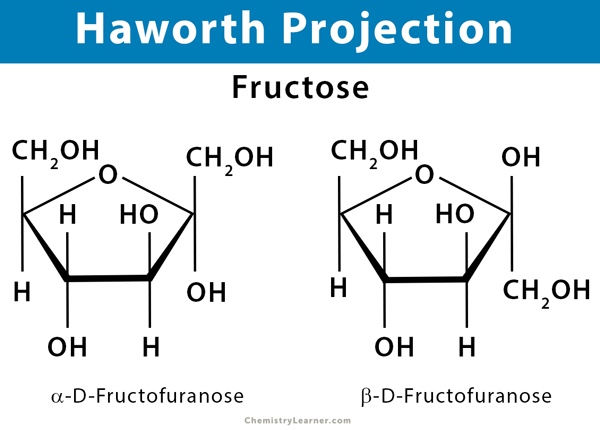
Six Carbon Ketone or Ketohexose: Psicose, fructose, sorbose, and tagatose
Disaccharide
12 Carbon Atoms: Sucrose, lactose, and maltose
How to Draw a Hayworth Projection?
Haworth projection depicts a pyranose as a hexagon (6-membered ring) and a furanose as a pentagon (5-membered ring). The substituent groups are placed straight up and down. A flat polygon is drawn to represent the ring. The oxygen in the ring is placed at the upper right in pyranose and upper center in furanose. Usually, the hemiacetal carbon at the anomeric position is placed at the extreme right. The thicker bonds between carbon atoms at the bottom of the ring are closer to the viewer. The thinner bonds at the top of the ring are further away. The vertical bonds attached to the ring are used to indicate substituents above and below the ring. The hydrogen and hydroxyl bonds below the ring represent those on the right in a Fischer projection.
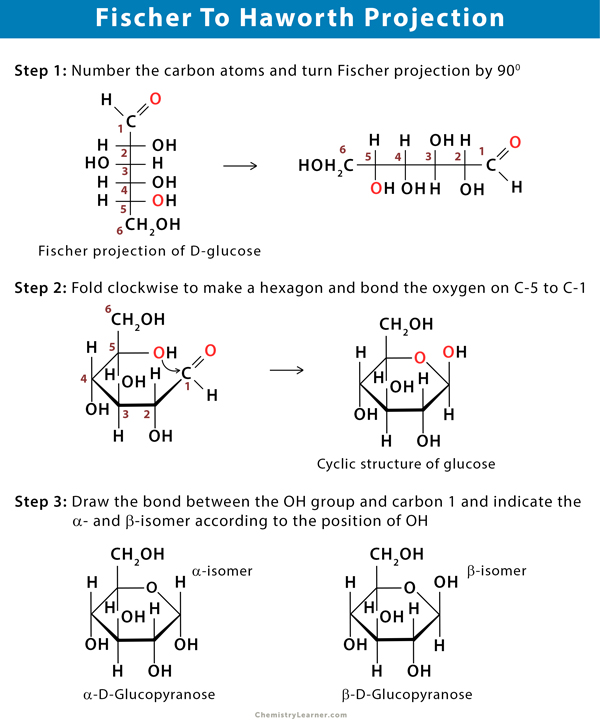
Conversion of Fischer Projection to Haworth Projection
The conversion of Fischer projection to Haworth projection involves a few sequences of steps.