Hydrohalogenation
What is Hydrohalogenation (2)
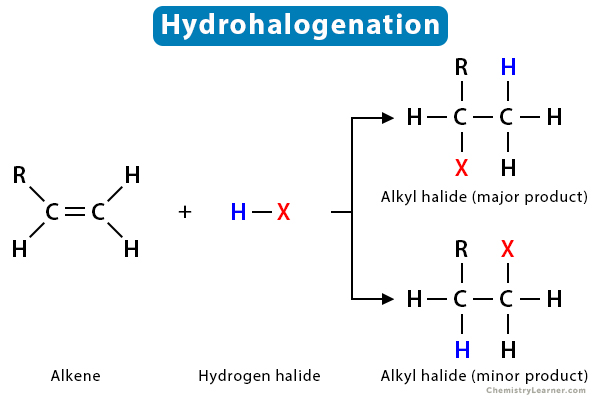
Hydrohalogenation is a type of electrophilic addition reaction that occurs in unsaturated compounds like alkene and alkyne. A hydrogen halide (HX), such as hydrogen chloride (HCl) or hydrogen bromide (HBr), adds to an alkene, producing the corresponding haloalkanes or alkyl halides. The reaction is carefully carried out in the presence of a non-polar or inert solvent like carbon tetrachloride (CCl4). The pi bonds in alkenes and alkynes are easily susceptible to electrophilic attack.
General Reaction
To understand the fundamental reaction of hydrohalogenation, let us take an alkene R1HC=CH2, which reacts with a hydrogen halide HX (X = Cl, Br, F, I). The reaction yields two alkyl halides – R1HXC-CH3 as the major product and R1H2C-CH2X as the minor product. The reaction is as follows:
R1HC=CH2 + HX → R1HXC-CH3 (major) + R1H2C-CH2X (minor)
Hydrohalogenation of Alkenes (1-3)
Examples
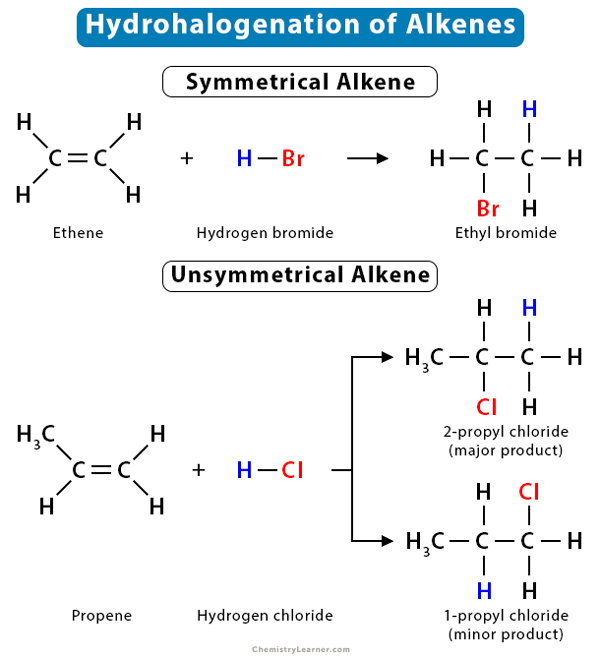
1. Reaction of Ethene (CH2=CH2) with HBr
Ethene is a symmetrical alkene containing two carbon atoms. It reacts with HBr in the presence of CCl4, producing bromoethane or ethyl bromide (CH3CH2Br).
CH2=CH2 + HBr → CH3CH2Br
2. Reaction of Propene (CH3CH=CH2) with HCl
Propene is an unsaturated alkene consisting of a three-carbon chain with two carbon atoms on either side of the double bond. When it reacts with hydrogen chloride, propene produces 2-chloropropane or 2-propyl chloride (CH3CHClCH3) as the major product and 1-chloropropane (CH3CH2CH2Cl) as the minor product.
CH3CH=CH2 + HCl → CH3CHClCH3 (major product, more stable)
+
CH3CH2CH2Cl (minor product, less stable)
Here, CH3CHClCH3 and CH3CH2CH2Cl are constitutional isomers of each other.
Mechanism
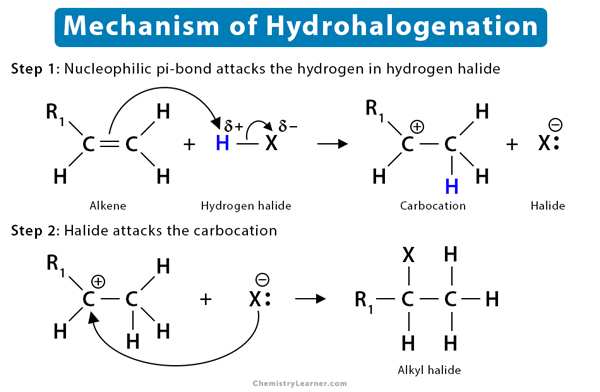
The fundamental mechanism of hydrohalogenation of alkenes is as follows:
- First, the nucleophilic pi bond of the alkene breaks down as it tries to grab the electrophilic H+ from HX.
- Then, Markovnikov’s rule is applied. H+ adds to the less substituted carbon atom. The more substituted carbon is now deficient in electrons, thus turning into a carbocation.
- Finally, the halide ion (X–) present in the solution attacks the carbocation forming a haloalkane or alkyl halide.
To understand the mechanism of hydrohalogenation of alkenes in-depth, let us go through each step in detail:
Step 1: Use an Inert Solvent
An inert or non-polar solvent, such as dichloromethane (CH2Cl2) and carbon tetrachloride (CCl4), dissolves the reactants and reagents. However, it does not interfere in the reaction process. If any non-inert or polar solvent is used for the reaction, the result will be completely different. For instance, if carried out in water, the final product will be alcohol. On the other hand, if carried out in an alcoholic medium, the product will be ether.
Step 2: Understand the Nature of the Molecules
Now, try to understand the nature of the reacting molecules. Here, the reactants are an alkene and a hydrogen halide (HX). HX molecules are highly polar since halogens like Cl, Br, I, and F are highly electronegative. Due to this, the halogen becomes partially negative (δ–). It pulls the electrons away from hydrogen, making it partially positive (δ+). So, hydrogen halide provides both an electrophile (H+) and a nucleophile (X–).
[Note – HF seldom shows this reaction due to fluorine’s high electronegativity and poor reactivity.]
Step 3: Break the Pi(π) Bond
In this step, the alkene’s pi (π) bond reaches out to grab the electrophilic hydrogen atom. For this, a pi (π) electron must let go of the carbon atom that it is bound to, yielding a carbocation. The carbocation is a high-energy intermediate, making this the reaction’s rate-determining step (slow step).
Markovnikov’s Rule and Regioselectivity
The next thing that comes to our mind is, which carbon atom of the double bond will take up the hydrogen? According to Markovnikov’s rule, the carbon with fewer substitutions in a double bond of an unsymmetrical alkene will first grab the H+, producing a carbocation. In other words, the carbon with more hydrogen atoms attached to it will take the H+. So, the more substituted carbon atom of the double bond is converted to a stable carbocation, an electrophile. It means that the reaction is regioselective.
Rearrangement of Carbocation
Generally, the formation of carbocation follows Markovnikov’s rule. However, there is a chance that the carbocation rearranges by shifting the hydride (H–) or methyl (CH3–) group and result in more than one stable carbocation. The stability of carbocation is as follows: 3° > 2° > 1°. The reaction will always prefer the 2° or 3° carbocation as it is more stable than 1°. So, Markovnikov’s rule does not hold for every case, as it does not deal with carbocation rearrangement.
Example of Carbocation Rearrangement
When 3-methyl-1-butene (CH3CH(CH3)CH=CH2) reacts with hydrogen bromide (HBr), the major product will be 3-methyl-2-bromobutane CH3CH(CH3)CH(Br)-CH3.
CH3CH(CH3)CH=CH2 + HBr → CH3CH(CH3)CH(Br)-CH3
However, there is a probability of producing 3° carbocation by 1,2-hydride shifting. As we know that 3° carbocation is more stable than 2°, this rearrangement will produce a more stable alkyl halide.
Step 1: Formation of 2° carbocation
CH3CH(CH3)CH=CH2 + HBr → CH3CH(CH3)C+HCH3
Step 2: Rearrangement by 1,2-hydride shift to form 3° carbocation
CH3CH(CH3)C+HCH3 → CH3C+(CH3)CH2-CH3
Step 3: Addition of bromide (Br–)
CH3C+(CH3)CH2-CH3 + Br– → CH3CBr(CH3)CH2-CH3
Anti-Markovnikov’s Rule
Sometimes, H+ may bind to a more substituted carbon rather than the least substituted carbon, following anti-Markovnikov’s rule. This process is quite unusual, as carbocations formed in the majority during hydrohalogenation reactions tend to favor the more substituted carbon.
Step 4: Attack of the Halide
In this step, the nucleophilic halide ion (X–) attacks the electrophilic carbocation. The halogen uses one of its lone electron pairs to attack the carbocation forming a sigma bond. In this context, stereochemistry is essential in determining the final product.
Stereochemistry
The nucleophile may attack the carbocation from either direction – top or bottom of the pi-bond. So, a mixture of syn (adding the substitutes to the same side of the pi-bond) and anti-products (adding the substitutes to the opposite sides of the pi-bond) can be expected here.
Hydrohalogenation of Alkynes (4-5)
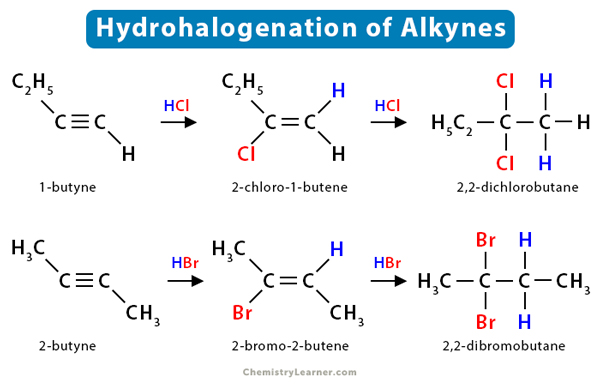
Like alkenes, alkynes also undergo hydrohalogenation reactions. Upon adding HX, alkynes produce a vinyl halide. This alkene is later converted into a geminal dihalide upon the second electrophilic addition of HX.
Like alkenes, the process starts by adding H+ to the π bond, forming a vinyl carbocation. The reaction is performed in the dark in a peroxide-free solvent. The more stable 2° carbocation is then attacked by the halide ion (X–), forming vinyl halide. The resulting haloalkene is less reactive since the electronegative halogen decreases the electron density of the double bond by the inductive effect, i.e., pulling the negative charge of the double bond towards itself. The reaction can be stopped at this point.
If further HX is added, another halide ion (X–) gets attached to the same carbocation, yielding germinal dihalide. It is an organic compound containing two halide groups attached to the same carbon atom.
General Reactions
Step 1: Formation of 2° Vinyl Carbocation
RC≡CH + HX → RC+=CH2
Step 2: Formation of vinyl halide
RC+=CH2 + X– → RCX=CH2
Step 3: Formation of 2° Carbocation
RC(X)=CH2 + HX → RC+(X)-CH3
Step 4: Formation of Geminal Dihalide
RC+(X)-CH3+ Cl– → RC(X)(X)-CH3
Example
Consider the reaction of propyne (CH3-C ≡C-H) with hydrogen bromide (HBr).
Step 1: Formation of 2° vinyl carbocation
CH3-C ≡C-H + HBr → CH3C+=CH2
Step 2: Formation of 2-bromopropene
CH3C+=CH2 + Br– → CH3CBr=CH2
Step 3: Formation of 2° carbocation
CH3CBr=CH2 + HBr → CH3C+Br-CH3
Step 4: Formation of 2,2-dibromopropane
CH3C+Br-CH3 + Br– → CH3C(Br)(Br)-CH3
Practice Problems
P.1. Mention the major product formed for the following reaction: Addition of HCl to 3,3-dimethyl-1-butene.
Sol. Following Markovnikov’s rule, 3,3-dimethyl-1-butene (CH3-C(CH3)(CH3)CH=CH2) first forms a 2° carbocation, which later gets attacked by Cl– to form 3,3 dimethyl-2-bromobutane (CH3-C(CH3)(CH3)CHBr-CH3).
The reaction is as follows:
CH3-C(CH3)(CH3)CH=CH2 + HBr → CH3-C(CH3)(CH3)C+H-CH3 → CH3-C(CH3)(CH3)CHBr-CH3
There is also a possibility to produce a 3° carbocation by shifting the adjacent methyl group.
Step 1:
CH3-C(CH3)(CH3)CH=CH2 + HBr → CH3C+CH3-CHCH3(CH3) + Br–
Step 2:
CH3C+CH3-CH(CH3)(CH3) + Br– → CH3CBrCH3-CHCH3(CH3)
As 3° carbocation is more stable than 2° carbocation, the second mechanism will be preferred.
P.2. What happens when acetylene is treated with HBr?
Sol. When acetylene (CH≡CH) is treated with HBr, it forms ethylidene bromide (CH3CHBr2). The reaction is as follows:
Step 1:
HC≡CH + HBr → H2C=C+H + Br– → CH2=CHBr
Step 2:
H2C=CHBr + HBr → H3C-C+HBr + Br– → H3C-CHBr2
References- Alkene Reactions – Masterorganicchemistry.com
- Hydrohalogenation Of Alkenes – Reaction Mechanism – Leah4sci.com
- Hydrohalogenation—Electrophilic Addition of HX – Chem.libretexts.org
- Reactions of Alkynes – Addition of HX and X₂ – Chem.libretexts.org
- Electrophilic Addition to Alkynes: Hydrohalogenation – Jove.com