Lewis Acid and Base
Definition: What are Lewis Acid and Lewis Base?
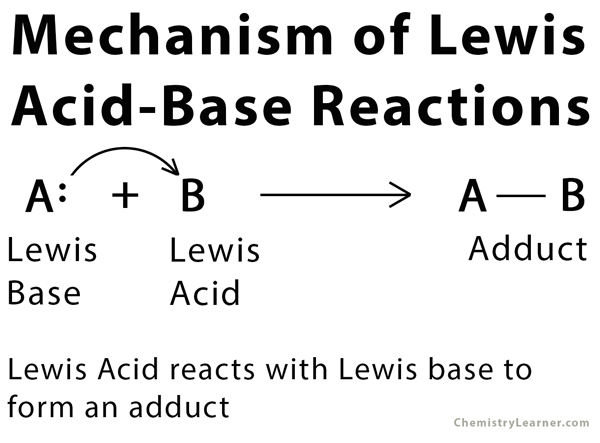
A Lewis acid is an electrophilic species that accepts a pair of electrons from ions or molecules. On the contrary, a Lewis base is a nucleophilic species that donates a pair of electrons to ions and molecules. Lewis acid and base can react with one another by transferring electrons to form a covalent bond resulting in a simple or complex molecular structure. The product formed by a Lewis acid-base reaction is known as adduct [1 – 4].
Mechanism of Lewis Acid-Base Reaction
Lewis Dot Structure
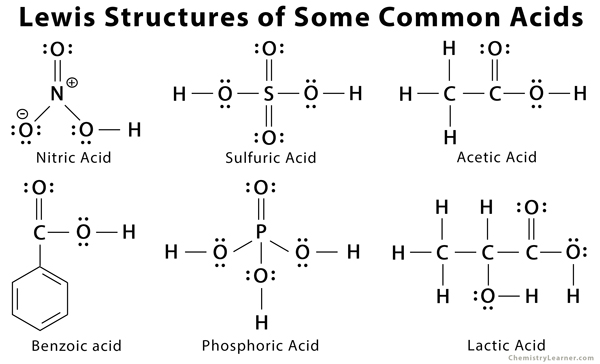
A Lewis dot structure is a straightforward representation of valence shell electrons in an ion or molecule. It shows how electrons are positioned around the atoms in a chemical species. The paired electrons are represented by dots, and the bonded electrons are represented by a dash [9]. The Lewis dot structure of some common acids like nitric acid, sulfuric acid, acetic acid, benzoic acid, phosphoric acid, and lactic acid are shown in the figure below [5].
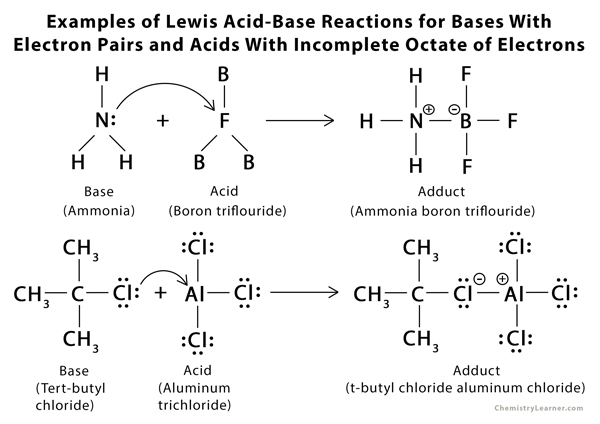
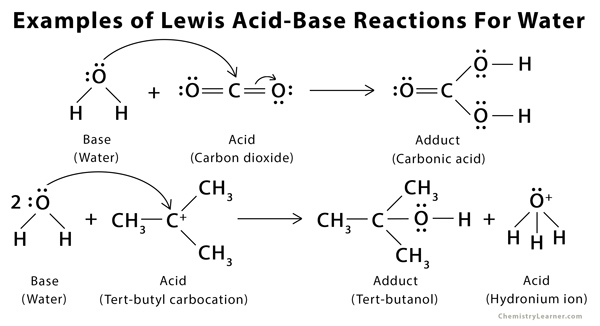
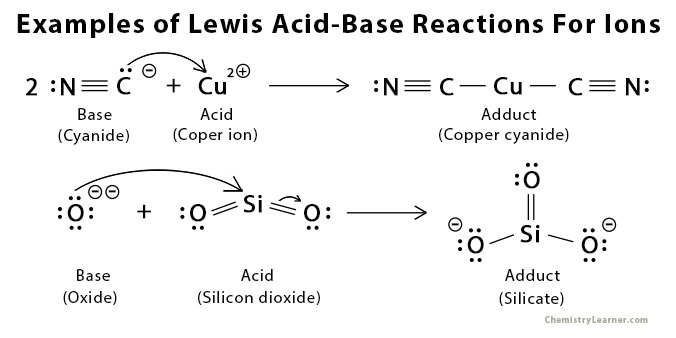
Examples of Lewis Acid and Base [6 – 9]
Lewis acid can be
- Any cation that can accept electron pair: H+, Mg2+, Fe3+, NH4+, and H3O+
- Any molecule with an incomplete octet of electrons: BF3 and AlCl3
- Any molecule where the central atom can have more than 8 electrons in its valence shell: SiCl4 and SiF4
- Any molecule that can have multiple bonds between two atoms of different electronegativity: CO2 and SO2
Lewis base can be
- Any simple anion that can give up a lone pair of electrons: O2-, Cl–, F–, and H–
- Any anionic species containing a lone pair of electrons: OH–, CN–, CH3–, and SO42-
- Any molecule with an extra pair or pairs of electrons: H2O, NH3, and C(CH3)Cl
- Any electron-rich pi system: benzene (C6H6), ethylene (C2H4), and ethyne (C2H2)
Lewis vs. Bronsted Theory
According to Lewis theory, an acid is an acceptor of electrons, and a base is a donor of electrons. According to Bronsted theory, an acid is a proton (H+ ion) donor, and a base is a proton acceptor [10].
Applications of Lewis Acid [11]
- Friedel-Crafts alkylation reaction
Applications of Lewis Base [11]
- Formation of ligands
- Modification of metallic catalyst
- Form bonds with Lewis acids to produce chelating agents
FAQs
Q.1. Why is acetic acid not a Lewis acid?
Ans. Acetic acid is not a Lewis acid because it cannot form a covalent bond with an electron pair.
Q.2. Is CH4 a Lewis acid or base?
Ans. It is neither a Lewis acid nor a base. CH4 is a non-polar molecule with a low electronegativity difference between C and H.
Q.3. Is iodine a Lewis acid or base?
Ans. Iodine is a Lewis acid because it can combine with Lewis base to form complexes. For example, it gives a red solution with benzene.
Q.4. Is CO2 a Lewis acid or base?
Ans. CO2 is a Lewis acid since it has a slightly positive charge on the C atom, due to significant electronegativity difference with oxygen, and can attract electrons.
Q.4. How to identify Lewis acid and bases?
Ans. The best way to identify if an ion or a molecule is a Lewis acid or base is by drawing its Lewis dot structure. Then identify those electrons that are bonded and the ones that are not. The electronegativity difference between the bonded atoms and the presence of pi electrons should also be taken into account.
- References
- Definition – Study.com
- Definition –Chem.libretexts.org
- Definition – Chemed.chem.purdue.edu
- Definition – Chemicool.com
- Structure – Chem.libretexts.org
- Examples – Opentextbc.ca
- Examples – Chem.ucla.edu
- Examples – Britannica.com
- Examples –Meta-synthesis.com
- Lewis Acid vs Bronsted Acid – Chem.libretexts.org
- Applications – Byjus.com





Thank you