Limiting Reagent (Reactant)
What is Limiting Reagent [1 – 4]
A limiting reagent or limiting reactant is a substance that has been wholly consumed in a chemical reaction. Thus, the limiting reagent determines when to complete and stop a reaction. Since the limiting reagent is consumed in a reaction, no amount remains to react with another reactant. Therefore, the other reactant remains in excess and is known as the excess reagent. The amount of products resulting from the chemical reaction is limited by the limiting reagent.
Limiting reagent is important in chemistry because it tells a chemist how much product will be produced if a limited amount of reactants are used in the reaction.
Examples of Limiting Reagent [1 – 5]
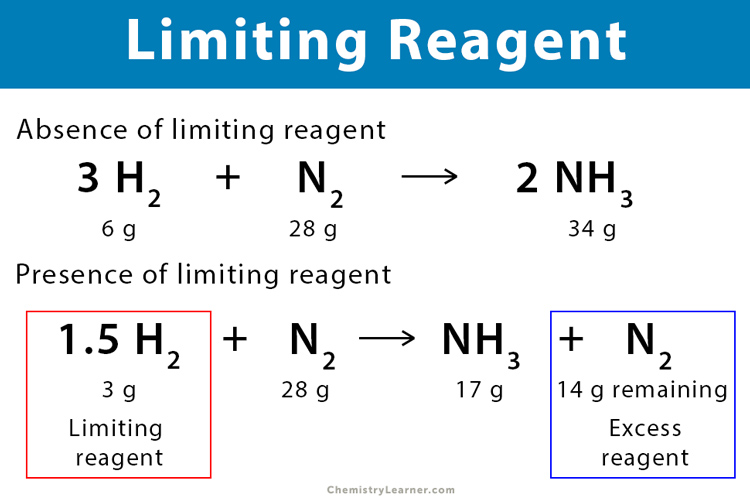
Example 1: Consider the reaction between hydrogen (H2) and nitrogen (N2) to form ammonia (NH3). In this reaction, three moles of H2 react with one mole of N2 to produce two moles of NH3. The balanced chemical reaction is given as follows:
3 H2 + N2 → 2 NH3
This relationship among the different quantities or moles of the reactants and products is known as stoichiometry. In the above reaction, H2, N2, and NH3 are in stoichiometric amounts.
If two moles of H2 react with one mole of N2, then all N2 will not be used since there is insufficient H2 in the reaction. Therefore, hydrogen is the limiting reagent.
Example 2: Consider the reaction between two moles of H2 and one mole of oxygen (O2) to give two moles of water (H2O).
2 H2 + O2 → 2 H2O
If instead, one mole of H2 reacts with one mole of O2, the reaction will not be complete, and hydrogen becomes the limiting reagent.
How to Find Limiting Reagent [1 – 4]
When two reactants mix to form products, they combine in a specific ratio determined by the reaction’s stoichiometry. The first step is to balance the chemical reaction. Consider the following balanced reaction:
A + B → Products
Suppose the quantities of A and B are known in a mass unit, such as grams. The next step is to convert grams to moles.
Formula to Calculate the Number of Moles from Mass
To find the concentrations of the reactants and products in moles, one has to know their quantities and molar masses. Suppose x grams of A, whose molar mass is M grams, are present in the reaction. Then, the amount in moles n is given by:
n = x/M
There are two general methods to find the limiting reagent.
Method 1
One trick to determine the limiting reagent is to compare the reactants’ molar ratio to the actual stoichiometric ratio. The reactant having fewer moles than required is the limiting reagent. Then, proceed to calculate the amount of product produced by the reactants and the remaining excess reagent left in the reaction.
Method 2
Another method of determining the limiting reagent is comparing the amounts of products formed from each reactant. An advantage of this method over the previous method is that it can be extended to many reactants. After balancing the chemical reaction, use stoichiometry to calculate the quantities of the products produced for each separate reactant. The reactant that produces the least amount of products is the limiting reagent. The amount of the products in grams formed from the limiting reagent is defined as the theoretical yield.
Example: Consider the reaction between solid aluminum (Al) and gaseous chlorine (Cl2) to give sodium aluminum chloride (AlCl3). The reaction is as follows:
2 Al (s) + 3 Cl2 (g) → 2 AlCl3 (s)
Suppose 3.5 grams of Al and 5.2 grams of Cl2 are present in the reaction. Determine the limiting reagent. Also, calculate the remaining amount of excess reagent and the amount of product produced. The molar mass of Al is 27 g, and that of Cl2 is 71 g.
Solution:
Step 1: Calculate the number of moles of Al and Cl2.
Number of moles of Al = 3.5 g/27 g = 0.13 mol.
Number of moles of Cl2 = 5.2 g/71 g = 0.073 mol.
Using Method 1
Step 2: Calculate and compare the required and actual molar ratios.
Molar ratio = Number of mol. of Al/Number of mol. of Cl2
Required molar ratio = 2/3 = 0.66
Actual molar ratio = 0.13/0.073 = 1.775
The actual molar ratio of Al to Cl2 is higher than required, which means that Al is in excess. If Al is excess, Cl2 must be in a limited amount. Therefore, Cl2 is the limiting reagent.
Using Method 2
Step 2: Calculate the amount of AlCl3 produced from the given amounts of Al and Cl2.
2 mol. of Al produces 2 mol. of AlCl3
Therefore, 0.13 mol. will produce = 2/2 x 0.13 = 0.13 mol. of AlCl3
3 mol. of Cl2 produces 2 mol. of AlCl3
Therefore, 0.073 mol. will produce = 2/3 x 0.073 = 0.049 mol. of AlCl3
Since Cl2 produces the least amount of AlCl3, it is the limiting reagent.
The molar mass of AlCl3 is 27 + 3/2 x 71 = 133.5 g
Therefore, amount of AlCl3 produced in the reaction = 0.049 x 133.5 = 6.54 g.
Step 3: Calculate the remaining amount of excess reagent.
Al is the excess reagent.
3 mol. of Cl2 reacts with 2 mol. of Al
Therefore, 0.073 mol. of Cl2 reacts with 2/3 x 0.073 = 0.049 mol. of Al
Amount of Al present = 0.13 mol.
Therefore, remaining amount of Al = 0.13 – 0.049 = 0.081 mol.
Remaining amount of Al in gram = 0.081 x 27 = 2.2 g
Solved Problems
P.1. Consider the following reaction:
2 Na + Cl2 → 2 NaCl
3.5 g of sodium (Na) metal is transferred to a 4L flask filled with chlorine (Cl2) gas at STP. Determine the limiting reagent and the remaining amount of excess reactant present if the molar mass of Na is 23 g, the molar mass of Cl2 is 71 g, and the molar volume of Cl2 is 22.4 L.
Solution.
Step 1: Determine the number of moles.
Number of mol. of Na = 3.5/23 = 0.152 mol.
Number of mol. of Cl2 = 4/22.4 = 0.179 mol.
Step 2: Using method 1 to calculate the molar ratios.
Molar ratio = No. of mol. of Na/No. of mol. of Cl2
Required molar ratio: 2/1 = 2
Actual molar ratio: 0.152/0.179 = 0.85
The actual molar ratio is less than required, which means that Na is the limiting reagent.
Step 3: Determine the remaining amount of excess reagent.
The excess reagent is Cl2.
2 mol. of Na react with 1 mol. of Cl2
Therefore, 0.152 mol. of Na react with = ½ x 0.152 = 0.076 mol. of Cl2
Amount of Cl2 present in the reaction = 0.179 mol.
Therefore, remaining amount of Cl2 = 0.179 – 0.076 = 0.103 mol.
Remaining amount of Cl2 in L = 0.103 x 22.4 = 2.3 L
P.2. Identify the limiting reactant if 4.45 moles of sodium (Na) react with 3.75 moles of oxygen (O2) in the following equation:
4 Na + O2 → 2 Na2O
Also, find the remaining amount of excess reagent and the amount of product produced.
Solution.
Step 1: Find the limiting reagent using method 2.
4 mol. of Na produces 2 mol. of Na2O
Therefore, 4.45 mol. of Na produces 2/4 x 4.45 = 2.23 mol. of Na2O
1 mol. of O2 produces 2 mol. Na2O
Therefore, 3.75 mol. of O2 produces 2/1 x 3.75 = 7.5 mol. of Na2O
In the above reaction, Na produces less amount of Na2O than O2. Therefore, Na is the limiting reagent.
The amount of Na2O produced in this reaction is 2.23 mol.
Step 2: Calculate the remaining amount of excess reagent.
O2 is the excess reagent.
4 mol. of Na react with 1 mol. of O2
Therefore, 4.45 mol. of Na react with ¼ x 4.45 = 1.11 mol. of O2
Hence, the remaining amount of O2 is 3.75 – 1.11 = 2.64 mol.