Excess Reagent
What is an Excess Reagent (1-5)
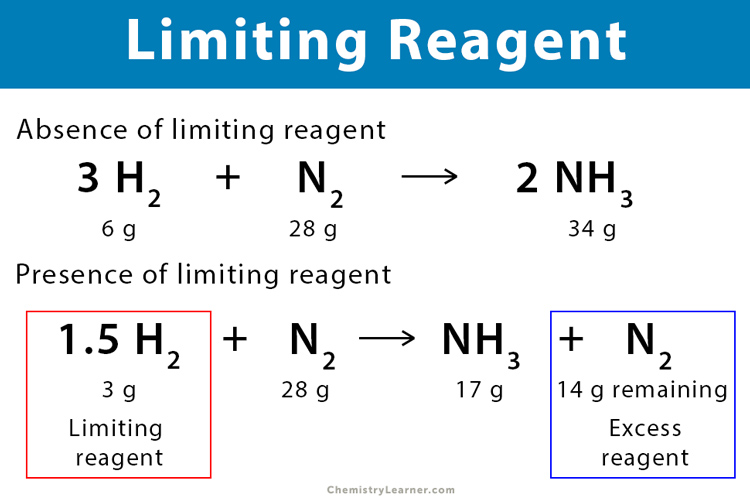
Excess reagent is the reactant or reagent that is left after a chemical reaction is complete. In other words, when a chemical reaction takes place, a reactant remains present in greater quantity than required to complete the reaction. It is not entirely consumed during a reaction. Because of its presence in excess amount, it is referred to as the excess reagent.
On the other hand, some reactants react wholly and get consumed within a reaction. This type of reactant is known as a limiting reagent.
Examples of Excess Reagent (1, 3)
Example 1: Silver iodide (AgI) reacts with sodium sulfide (Na2S) to produce silver sulfide (Ag2S) and sodium iodide (NaI).
AgI + Na2S → Ag2S + NaI
The balanced reaction is:
2 AgI + Na2S → Ag2S + 2 NaI
From the balanced chemical equation, it is evident that there is a 2:1 mole ratio between silver iodide and sodium sulfide. Suppose 1 mole of each reactant is used for the reaction. In that case, the silver iodide will get consumed totally, and sodium sulfide will remain in excess. So, the reaction will stop, as the silver iodide will be finished.
Excess reagent: Na2S
Limiting reagent: AgI
Example 2: Magnesium hydroxide [Mg(OH)2] reacts with hydrochloric acid (HCl), yielding magnesium chloride (MgCl2) and water(H2O).
Mg(OH)2 + HCl → MgCl2 + H2O
The balanced reaction is:
Mg(OH)2 + 2 HCl → MgCl2 + 2 H2O
From the balanced chemical equation, we can observe a 1:2 mole ratio between magnesium hydroxide and hydrochloric acid. Suppose 1 mole of each reactant is used for the reaction. In that case, the hydrochloric acid will get used up totally, and magnesium hydroxide will remain in excess. So, the reaction will cease after a while, as the hydrochloric acid will be finished.
Excess reagent: Mg(OH)2
Limiting reagent: HCl
How to Find Excess Reagent (3-5)
To understand how to find an excess reagent in a chemical reaction, let us take the example of the combustion of methane (CH4).
The reaction is:
CH4 + O2 → CO2 + H2O
Step 1: Balance the chemical reaction.
To calculate the amount of excess reagent, first, we need to balance the chemical reaction.
Balanced chemical reaction:
CH4 + 2 O2 → CO2 + 2 H2O
Step 2: Calculate the molecular mass of each reactant.
To find out the excess reagent, we need to deal with only the reactant side. Next, calculate the molecular mass of each reactant.
For the given reaction,
Molecular mass of CH4 = 12 + (4×1) = 16 grams
Molecular mass of O2 = 2 x (16×2) = 64 grams
This 16:64 is a standard or fixed ratio for the combustion of methane. That means, for the combustion of 16 grams of methane, we will require 64 grams of oxygen.
Step 3: Identify the excess reactant and calculate the mass.
Now, let us assume we have only 8 grams of methane and 48 grams of oxygen available for the reaction. From the standard ratio, we already know that 64 grams of oxygen are required for combusting 16 grams of methane. Applying a simple unitary method, we can calculate how much oxygen will be required for combusting 8 grams of methane.
We know,
For 16g of CH4 → 64g of O2 is required
∴ For 8g of CH4 → [64/16) x 8]g = 32g of O2 is required
From the above calculation, it is clear that for the combustion of 8g of methane, 32g of oxygen is needed. However, we have 48g of oxygen present in the reaction. So, the extra oxygen will not get used up in the reaction, thus becoming the excess reagent.
Therefore, oxygen is the excess reagent here.
∴Amount of excess oxygen = [Total amount of O2 – Consumed O2] = 48g – 32g = 16g
During this process, the 8g of methane will be completely used up. Thus, CH4 is the limiting reagent in this reactant. The reaction will stop after this 8g of methane gets consumed totally.
References
- Overview of Excess Reactant in Chemistry –Thoughtco.com
- Excess and Limiting Reagents – Chem.libretexts.org
- Determining limiting and excess reagents – Uah.edu
- Limiting and excess reagents – Chemistryvce.weebly.com
- Moles, excess and limiting reagents – Studypug.com