Nucleophilic Addition
What is Nucleophilic Addition?
A nucleophile addition is an addition reaction in which a nucleophile adds to a pi bond of a compound. As a result, two new sigma bonds are formed. This reaction is essential in organic chemistry because it enables carbonyl compounds to form new products having various functional groups.
Carbonyl compounds undergo nucleophilic addition reactions because of the C=O bond’s polarity. The polar bond allows partial charges on the carbon and oxygen atoms, making it easy for the nucleophile to attack at the 1,2-positions.
Nucleophilic addition reaction will be most favored in aldehydes and ketones. It differs from nucleophilic substitution reactions since neither aldehydes nor ketones have a leaving group [1-7].
Reactivity of Carbonyl Compounds
The reactivity of carbonyl compounds towards nucleophilic addition reactions is influenced by the groups attached to the carbonyl carbon. The reactivity depends on the magnitude of the positive charge on the carbonyl atom and is high for electron-deficient carbon. The functional group, like alkyl, hinders the nucleophilic attack and decreases the electron deficiency and reactivity.
Example: The order of reactivity of formaldehyde (methanal) (HCHO), acetaldehyde (ethanal) (CH3CHO), and acetone ((CH3)2CO).
HCHO > CH3CHO > (CH3)2CO
Therefore, formaldehyde is most reactive toward nucleophilic addition reaction.
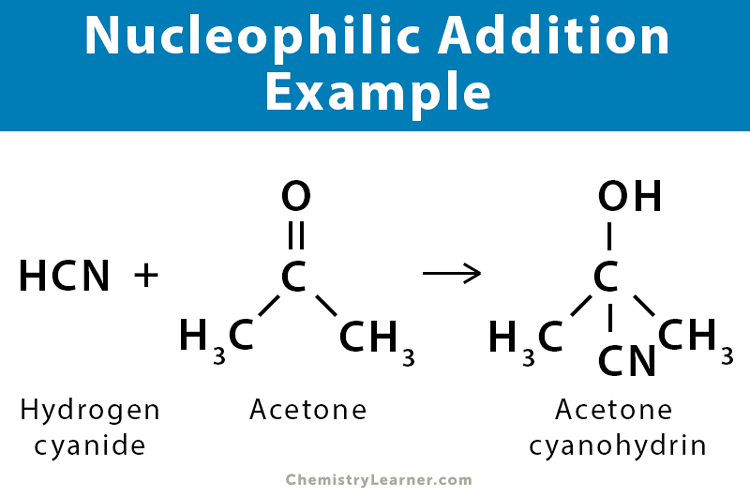
Examples of Nucleophilic Addition Reaction [1-5]
Here are some examples of the nucleophilic addition reaction.
1. Nucleophilic Addition of Water
The nucleophilic addition of water to a carbonyl compound like an aldehyde or ketone results in a geminal diol (hydrate). The reaction is usually slow under neutral conditions. However, the rate can be significantly increased through the addition of an acid or a base catalyst.
RCOR’ + H2O → RC(OH)2R’
Example: Acetaldehyde (CH3CHO) hydrolyzes to propane-2,2-diol (C3H8O2).
CH3CHO + H2O → C3H8O2
2. Nucleophilic Addition of Alcohol
Aldehydes and ketones undergo a nucleophilic addition reaction with alcohols to give hemiacetal, which reacts with another alcohol molecule to give acetal (geminal diethers). This reaction takes place in the presence of an acid catalyst.
RCOR’ + 2 R”OH → RC(OR”)2R’ + H2O
Example: Acetone (CH3COCH3) reacts with ethanol (EtOH) to give 2,2 diethoxy propane ((CH3)2C(OEt)2) and water (H2O).
CH3COCH3 + 2 EtOH → (CH3)2C(OEt)2 + H2O
Mechanism of Nucleophilic Addition
Here are the steps involved in the nucleophilic addition reaction of carbonyl compounds. An image of the mechanism can be found here.
Step 1: Nucleophile forms a bond with the electrophilic C=O carbon atom, resulting in rehybridization of the carbonyl carbon from sp2 to sp3. Electrons in the pi-bond are attracted to the electronegative oxygen atom resulting in a tetrahedral alkoxide intermediate.
Step 2: The alkoxide is protonated by the addition of an acid to form an alcohol.
Strong nucleophiles like RMgX (Grignard reagent), RLi, and RCCM add directly to the C=O bond, forming the intermediate. On the other hand, weak nucleophiles like H2O, ROH, and RNH2 require an acid catalyst [2-7].
FAQ
Ans. Carboxylic acid does not show nucleophilic addition reaction because of resonance. As a result, the lone pair of electrons on the oxygen are delocalized.