Nucleophilic Substitution
Definition: What is Nucleophilic Substitution?
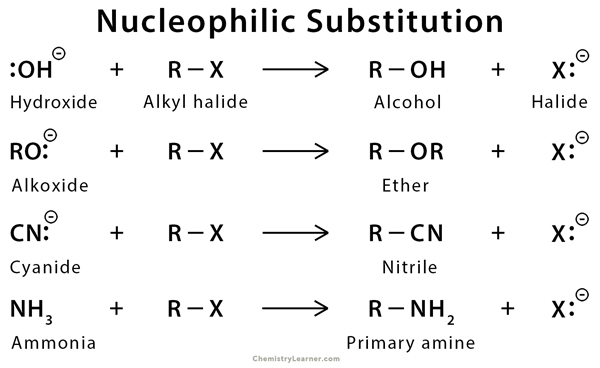
Nucleophiles, typically, have a lone pair of electrons in them. As the name suggests, they are attracted to the nucleus of an atom. Nucleophilic substitution is a substitution reaction in which an electron-rich nucleophile displaces the halogen atom bonded to the central carbon of an alkyl halide molecule. The halide ion that is displaced from the carbon atom is called the leaving group. Nucleophilic substitution reaction takes place at the site of a saturated carbon atom of aromatic, aliphatic, and acyl (RCO) compounds. Some typical nucleophiles are halide (X–), hydroxy group (OH–), alkoxy group (RO–), cyanide ion (CN–), hydrogen sulfide (H2S), ammonia (NH3), and water (H2O) [1-6].
The history of nucleophilic substitution reaction goes back to 1935, when British chemists Christopher Ingold and Edward D. Hughes first proposed a reaction mechanism.
Examples of Nucleophilic Substitution
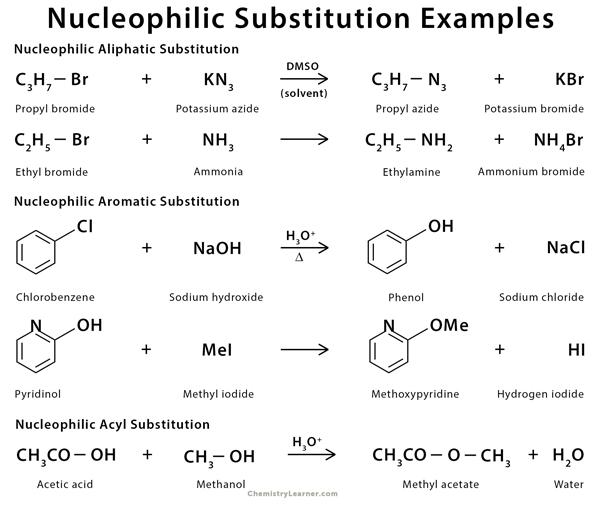
Nucleophilic substitution reaction can take place in aliphatic, aromatic, and acyl compounds. For example, propyl bromide, ethyl bromide, chlorobenzene, pyridinol, and acetic acid react with nucleophiles to give substituted compounds.
Principle of Nucleophilic Substitution
In alkyl and acyl halides, the bond between carbon and halogen is polar. The halogen being more electrophilic than the carbon, tends to attract the bonded electron pair. As a result, the halogen acquires a negative charge, and the carbon acquires a positive charge. Hence, the carbon becomes prone to a nucleophilic attack [1-6].
Types of Nucleophilic Substitution
There are two main types of nucleophilic substitution reactions – SN1 reaction and SN2 reaction.
SN1 Reaction
- Two-step reaction process: 1. carbon-halogen bond breaks, resulting in a positively charged carbon (carbonation), and 2. nucleophile attacks the carbocation, forming a new bond
- Mostly occurs in tertiary and secondary alkyl halides
- Unimolecular and follows first-order kinetics
- The speed of the reaction depends on the concentration of alkyl halide
- Single-step process, i.e., concerted reaction: nucleophile attacks the partially positive carbon atom that is bonded to the halogen, C-X bond breaks simultaneously, and the halogen leaves the molecule
- Mostly occurs in primary and secondary alkyl halides
- Bimolecular reaction and follows second-order kinetics
- The speed of the reaction depends on both the nucleophile and the haloalkane
SN2 Reaction
Difference Between SN1 and SN2 Reaction
SN1 vs. SN2 Reaction[7,8] | ||
| Property | SN1 | SN2 |
| Rate of reaction | Unimolecular, i.e., rate depends on the substrate | Bimolecular, i.e. rate depends on the substrate and nucleophile |
| Order of reactivity of haloalkane | 3°>2°>1° | methyl halide>1°>2° |
| Type of solvent | Polar protic (e.g., water and alcohol) | Polar aprotic (e.g., DMSO, acetone) |
| Strength of nucleophile | Weak | Strong |
| Stereochemistry | Racemization, i.e., a mix of retention and inversion | Inversion |
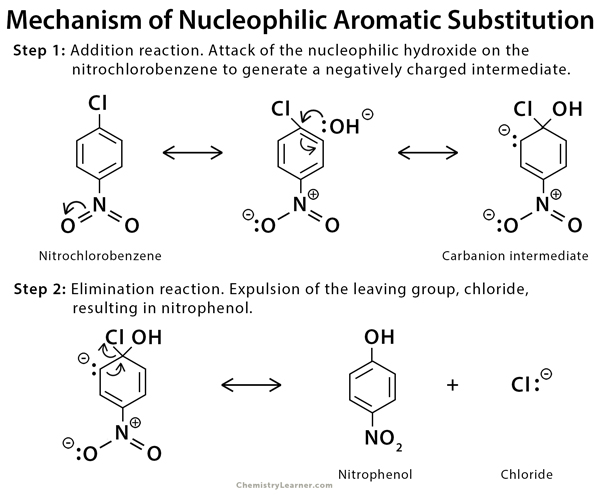
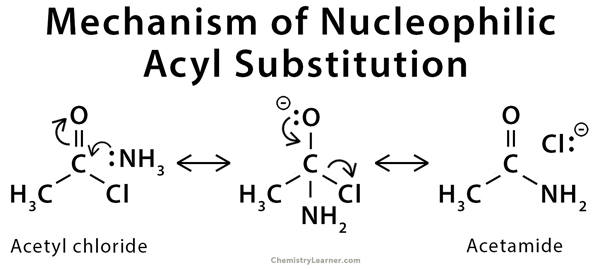
Mechanisms of Nucleophilic Substitution [9-13]
The mechanism of nucleophilic substitution reaction involves the addition of the nucleophile and the elimination of the leaving group.
FAQs
Ans. Chlorine is more electronegative than hydrogen and draws electrons in the ring towards itself. So, it reduces the electron density of the aromatic ring of the ring. Hence, chlorobenzene is less reactive than benzene in an electrophilic substitution reaction.
Ans. Vinyl chloride is less reactive than ethyl chloride towards nucleophilic substitution because of the double bond and sp2 hybridization, which results in the strengthening of the bond. Hence, a nucleophilic attack cannot result in substitution.
Ans. In aryl halides, the carbon-halogen bond has a partial double bond character. It becomes shorter, stronger, and challenging to break. Hence, it cannot be easily replaced by a nucleophile.
References
- Definition – Cliffsnotes.com
- Definition – Study.com
- Definition – Chemtube3d.com
- Definition – Chem.libretexts.org
- Definition – Masterorganicchemistry.com
- Definition – Chemguide.co.uk
- Comparison – Byjus.com
- Comparison – Masterorganicchemistry.com
- Mechanism – Chem.ucalgary.ca
- Mechanism – Chem.libretexts.org
- Mechanism – Organic-chemistry.org
- Mechanism – Masterorganicchemistry.com
- Mechanism – Cliffsnotes.com





like this topic
best site for chemist student