Primary, Secondary, and Tertiary Alcohols
Introduction
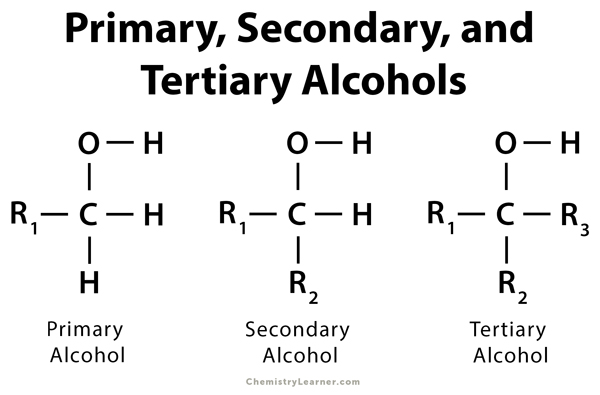
Alcohol is an organic compound that has a hydroxyl (-OH) group in its molecular structure. Each alcohol has an alpha-carbon to which the OH group is attached. Alcohol can be classified into three types depending upon the number of alkyl or aryl groups to which this alpha-carbon is bonded. They are called primary, secondary, and tertiary alcohols. The three different types of alcohol can be distinguished using the following principle [1-3].
Primary Alcohol
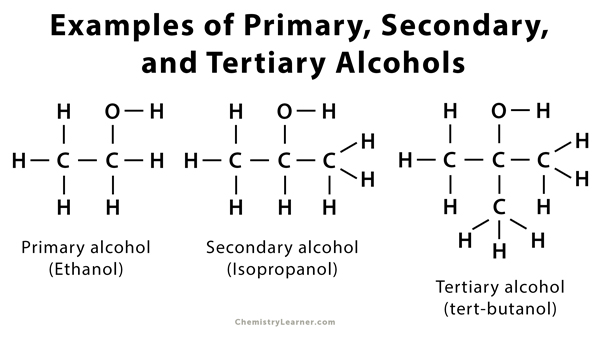
In the case of a primary alcohol, only one carbon atom is bonded to the alpha-carbon. Example – Ethanol, propanol, and butanol. If no carbon atom is bonded, then the primary alcohol is called methanol.
Secondary Alcohol
In the case of a secondary alcohol, two carbon atoms are bonded to the alpha-carbon. Example – 2 – propanol and 2 – butanol.
Tertiary Alcohol
In the case of tertiary alcohol, three carbon atoms are bonded to the alpha-carbon. Example –tert-butyl alcohol.
Preparation of Alcohols
There are many general methods of preparation of alcohol. These include [4-8]:
- Hydration of alkenes
- Hydroboration of alkenes
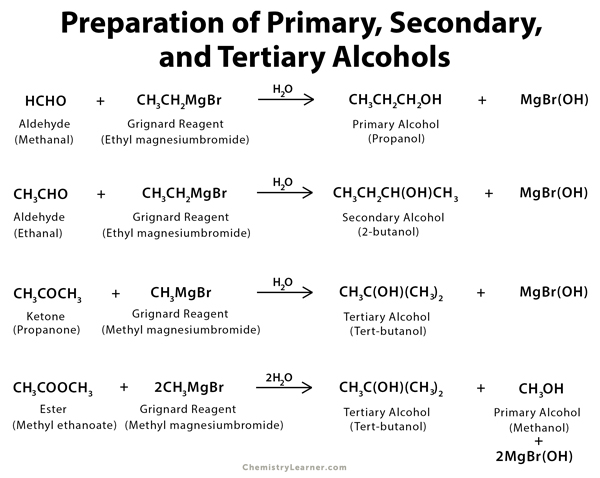
- Reaction of organometallic compounds with carbonyl compounds like aldehyde, ketone, and ester (see figure)
- Reaction of Grignard reagent with cyclic ethers
- Reduction of metal halide or boranes
- Catalytic hydrogenation of carbonyl compounds
- Meerwein–Ponndorf–Verley (MPV) reduction of aldehydes and ketones
- 1,2 glycols from alkenes
- Hydration of alkyl and allylic halides
- Hydrolysis of esters
- Aldol condensation
- Cleavage of ethers
Test for Distinguishing Alcohols
Lucas test is used to distinguish between primary, secondary, and tertiary alcohols. In this test, the reactivity of alcohols with Lucas reagent is tested, thus giving separate results for each type of alcohol.
Uses and Applications of Alcohols [9]
- Ethanol is used in spirits, as fuel, and as a solvent.
- Methanol is also used as fuel and in the manufacture of other organic compounds like methanal (formaldehyde), ethanoic acid, and methyl esters.
- Isopropanol is used as a solvent and a solution for cleaning surfaces.
- References
- Definition – Byjus.com
- Definition – Masterorganicchemistry.com
- Definition – Chem.libretexts.org
- Preparation – Askiitians.com
- Preparation – Chem.libretexts.org
- Preparation – Cliffsnotes.com
- Preparation – Jove.com
- Preparation – Toppr.com
- Application – Chemguide.co.uk





Nice answers, is the best ever.
well explained in simple terms. thank you for this blog
Thank you for those lovely answers you have to me, it was nice ????????????
I like chemistry