Aldol condensation
What is Aldol condensation?
It is the name given to an organic reaction of an enol or enolate ion with a carbonyl compound, resulting in the formation of a β-hydroxyketone or β-hydroxyaldehyde. This is succeeded by a dehydration that gives a conjugated enone.
The process is also simply known as Aldol reaction. It is an example of electrophilic replacement at the alpha carbon present in enolate or enols anions.
Aldol Condensation Name
The name of this process comes from the fusion of two words, “Aldol” and “Condensation”. The term “Aldol” refers to a colorless, oily liquid produced due to the condensation of two acetaldehyde molecules. The substance manufactured in this way consists of an aldehyde group (-CHO) and an alcohol group (-OH). The word “Condensation” means a reaction characterized by the combination of two molecules to produce a larger product accompanied by the loss of a water molecule.
A simple case of this reaction involves the combination of an aldehyde and an enolate to afford an alcohol. This is the reason behind the presence of “aldol” in Aldol condensation.
Aldol Condensation Reaction Mechanism
The reaction comprises of two parts. These are:
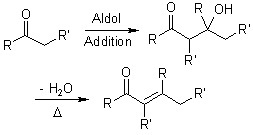
Picture 1 – Aldol condensation
Aldol reaction
This is the first part of the reaction which proceeds in three steps.
Step 1
This involves an acid-base reaction where hydroxide acts as a base and removes the acidic a-hydrogen thus producing the reactive enolate.
Step 2
In this step, the nucleophilic enolate reacts with the aldehyde present in the electrophilic carbonyl C in a process that leads to the development of an intermediate alkoxide.
Step 3
This comprises of an acid-base reaction in which a water molecule is deprotonated by the alkoxide thus forming hydroxide and the aldol products or b-hydroxyaldehydes.
Dehydration reaction
Also referred to as Elimination Reaction, this is the second part of the mechanism. The dehydration of the aldol addition product can occur through two mechanisms:
- In an enolate mechanism, through a strong base such as sodium hydride, potassium hydroxide or potassium t-butoxide.
- In an enol mechanism, catalyzed by acids.
In case of the presence of an activated carboxyl group Decarboxylation may accompany Dehydration. The products of aldol reactions often undergo a subsequent elimination of water, made up of an alpha-hydrogen and the beta-hydroxyl group.
Aldol Condensation Reaction
This reaction may occur within two molecules of the same ketone or the same aldehyde, or within different molecules (in a process known as “Crossed Aldol Reaction” or “Claisen-Schmidt Reaction”). It can occur in five ways:
Between two molecules of the same aldehyde
It is regarded as the classical case that involves only one aldol product. It may also involve an aldol product and the dehydrated product. The equilibrium of the reaction lies to the far right.
Between two molecules of the same ketone
The equilibrium lies to the left in this type of reaction. However, it can be shifted by continuous extraction or similar reaction conditions that are appropiate. In case of an unsymmetrical ketone, the condensation might take place on either side. This can produce a mixture of products.
In actual reactions, the condensation generally occurs on the side consisting of more hydrogens. The kinetic enolate is produced in this way.
Between two different aldehydes
Usually, such a reaction gives a mixture of four products. The count can go up to eight, if the dehydration products are also taken into account. In case one of the aldehydes does not have a α-hydrogen (such as benzaldehyde), the reaction can give only two products. In many cases, there is usually one main crossed product.
Between two different ketones
It is considered to be the toughest case, but can be carried out in case one of the ketones changes into a preformed enolate first.
Between an aldehyde and a ketone
Also known as the Claisen-Schmidt reaction, it is typically practicable due to the fact that aldehydes are stronger enolates compared to ketones. It is essential to use preformed enolates while using unsymmetrical ketones to insure regioselectivity.
If the product is dehydrated, problems like undesirable Michael Additions and Polycondensation may take place with the aldol reaction. The aldol reaction may be employed within the molecule to close five and six-membered rings, as observed in the Robinson annulation reaction.
Aldol condensation reaction can be catalyzed by either acids or bases though base catalysts are usually more preferred. This reaction was earlier supposed to take place only with aldehydes. It has, however, now been understood that a similar reaction would occur with reactive carbonyl compounds and ketones with available α-hydrogens.
Mixed Aldol Condensation
The process is also known as “Crossed Aldol Condensation” and occurs during the combination of two different molecules that contain carbonyl groups. Generally, such a reaction is only practical if no α-hydrogens are present in one of the compounds. This way, only one nucleophile is formed and only a single enol or enolate is generated.
Typically, a mixed reaction occurs between a ketone and an aldehyde having no α-hydrogens. This leads to the nucleophile being generated entirely from the ketone. The aldehyde is usually more reactive towards nucleophiles compared to the ketone. This further reduces the possibility of unwanted self-condensation of the ketone.
Application of Aldol Condensation
The reaction allows the formation of carbon-carbon bonds. In Gluconeogenesis and Photosynthesis, the reaction leads to the establishment of a C-C bond. This is considered to be an important reaction in the biochemistry of metabolism, where it is the fifth step of Glycolysis. In Glycolysis, however, it works in an inverse manner and serves to terminate the carbon-carbon bond.
The reaction is commonly implemented to manufacture solvents, such as Isophorone and Diacetone alcohol. It works as an intermediate for the production of perfumes. It is also used in the manufacture of pharmaceuticals, such as a, b unsaturated ketones and the aromatic ketones known as chalcones. It is also generally used to create plasticizers.
Aldol Condensation is a classic reaction. It has been understood well for decades and is widely used even today. In fact, most reactions have fallen out of favor with passing time. The same does not hold true for this reaction. Its importance has remained the same if not greater with time.
- References
- http://en.wikipedia.org/wiki/Aldol_condensation
- http://www.organic-chemistry.org/namedreactions/aldol-condensation.shtm
- http://www.chem.ucalgary.ca/courses/350/Carey5th/Ch18/ch18-3-4.html
- https://chem.libretexts.org/Bookshelves/Organic_Chemistry/Supplemental_Modules_(Organic_Chemistry)/Reactions/Organic_Reactions/Aldol_Condensation




