Supercritical Fluid
Supercritical fluids are substances that exist above a critical point of pressure and temperature where no separate liquid and gas phases exist. However, the pressure is below what is required to compress it into a solid.
For example, the critical temperature of hydrogen is -239.85°C (33.3K), and the critical pressure is 12.96 bar (12.8 atm). Beyond this temperature and pressure, hydrogen reaches the supercritical fluid phase.
Baron Charles Cagniard de la Tour discovered supercritical fluids in 1822 while conducting experiments involving the discontinuities of the sound of a flint ball in a sealed cannon barrel filled with various fluids at various temperatures.
Properties
Supercritical fluids have properties between those of a gas and a liquid. Some general properties are:
- Low viscosity of a gas and high density of a liquid, thus cannot be liquefied. However, a gas can be converted to a liquid without crossing the boundary between the vapor and liquid phase just by lowering the temperature
- No surface tension as they are not subject to the vapor-liquid boundary
- Solubility in a supercritical fluid increases with the density of the fluid at a constant temperature
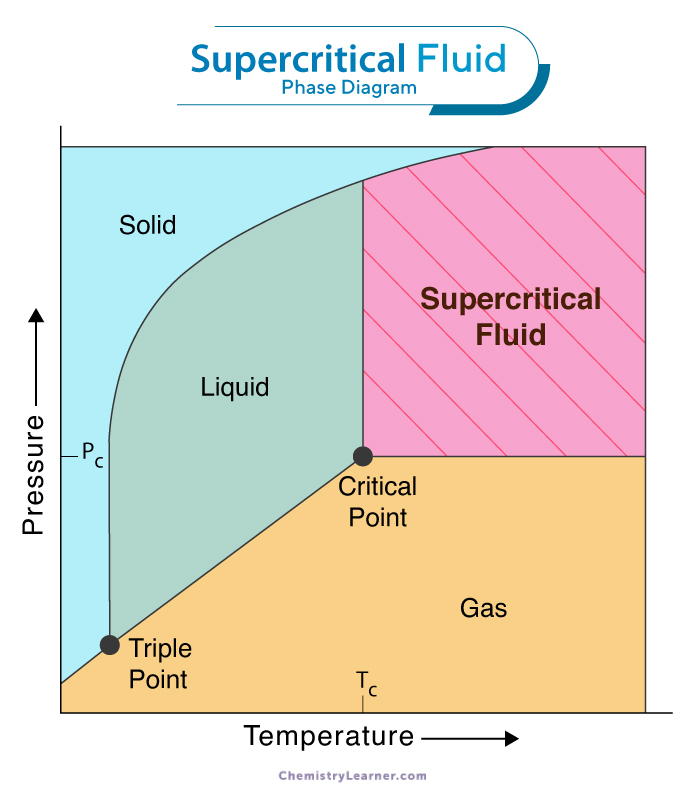
The concept of a supercritical fluid can be better understood using a pressure-temperature phase diagram.
Phase Diagrams
In a phase diagram, the lines of equilibrium denote a situation where two phases can coexist. The critical point is the maximum pressure and temperature at which liquid-gas equilibrium occurs. Beyond the critical point, such lines disappear as there is no difference between the liquid and gaseous phases, and the substance becomes supercritical. The phase diagram for fluids looks like the one given below.
Examples
The two most commonly used supercritical fluids are CO2 and water, the phase diagrams for which are illustrated below.
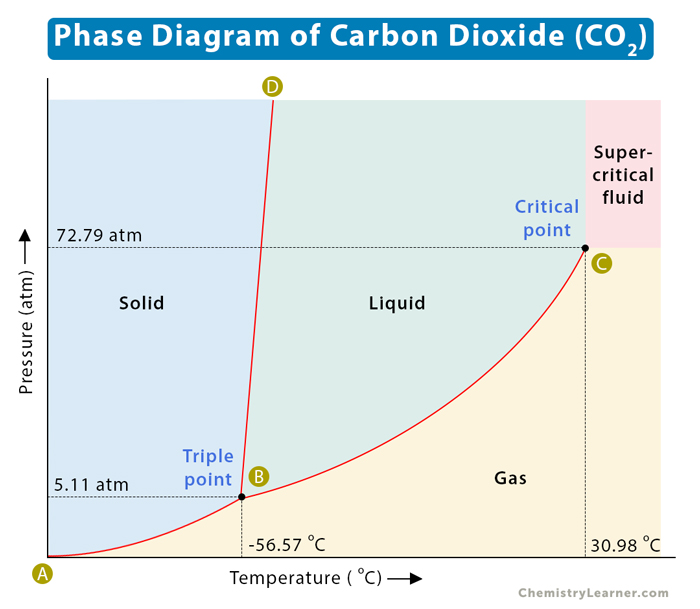
Carbon dioxide (CO2)
From the phase diagram, we observe that CO2 has a critical pressure of 73.8 bar (72.8 atm) and a critical temperature of 30.95°C (304K). Beyond this, it becomes a supercritical, non-toxic, non-flammable, and chemically inert fluid. Since it is cheaply made and highly pure, it is a versatile green solvent in many industries and processes. A notable use of supercritical CO2 is as an alternate solvent in extraction processes, as it does not leave a harmful organic residue.
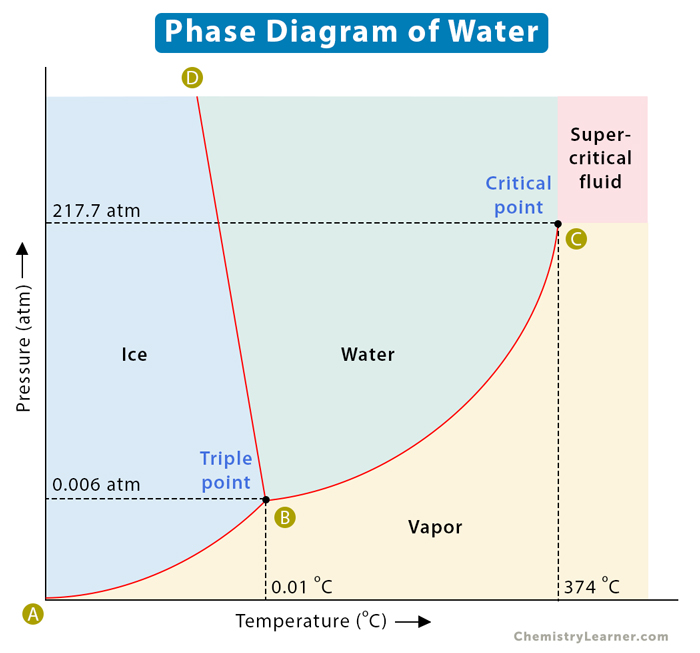
Water (H2O)
The critical pressure and temperature for water are 220.64 bar (217.775 atm) and 373.946°C (647.3K), respectively. Beyond this, it reaches the supercritical stage, as shown above. Unlike standard water, supercritical water is acidic and non-polar, so it finds use as a solvent in organic processes. It is also utilized in waste management as an oxidizer to neutralize harmful organic compounds.
The critical points of some more supercritical fluids are given:
| Solvent | Molecular mass (g/mol) | Critical temperature (C) | Critical pressure Bar (atm) | Critical density (g/cm3) |
|---|---|---|---|---|
| Carbon dioxide (CO2) | 44.01 | 30.95 | 73.8 (72.8) | 0.469 |
| Water (H2O) | 18.015 | 373.946 | 220.64 (217.755) | 0.322 |
| Methane (CH4) | 16.04 | -82.75 | 46.0 (45.4) | 0.162 |
| Ethane (C2H6) | 30.07 | 32.15 | 48.7 (48.1) | 0.203 |
| Methanol (CH3OH) | 32.04 | -241.11 | 80.9 (79.8) | 0.272 |
| Ethanol (C2H5OH) | 46.07 | 240.75 | 61.4 (60.6) | 0.276 |
| Acetone (C3H6O) | 58.08 | 234.95 | 47.0 (46.4) | 0.278 |
Supercritical fluids also exist in nature, such as underwater volcanoes and thermal vents. Here, high temperature and pressure cause the water to turn supercritical. Supercritical hydrogen and helium occur in the atmospheres of the gas giants, namely, Jupiter, Saturn, Uranus, and Neptune. Supercritical CO2 might exist in Venus’s atmosphere, but this is yet to be proven.
Applications
Supercritical fluids are employed in various industrial processes.
Supercritical Fluid Extraction
Supercritical fluid extraction (SFE) uses supercritical fluids to extract solvent while separating one component from another. SFE technology has advantages over liquid extraction due to its low viscosities, high solubility and diffusiveness, and the ability to tune the supercritical fluid for different components by varying temperature and pressure. Additionally, they are non-toxic, non-flammable, and environmentally safe. They are also simple to produce and can be retrieved by simple depressurization.
CO2 finds application in large-scale food industries such as the decaffeination of coffee beans, production of medicines and essential oils from plants, extraction of hops for beer manufacturing, and in laboratories for extraction instead of common solvents. The process requires pressurized equipment, and the CO2 is released back into the atmosphere after extraction.
Supercritical Fluid Chromatography
Supercritical fluid chromatography (SFC) is a form of normal phase chromatography. It works by using supercritical fluids as the mobile phase. The principle is similar to high-performance liquid chromatography (HPLC). It analyses and separates lightweight, thermally labile molecules and chiral compounds.
It is advantageous over HPLC and GC (gas chromatography) as it produces narrower peaks due to high diffusion rates, gives very pure results, and can be used with the universal flame ionization detector and non-volatile analytes. However, the cost of conducting SFC poses a disadvantage. Hence, it is mainly used in the pharmaceutical industry. It also purifies vegetable oils, polymers, plant extracts, and fish oil.
Sterilization
Supercritical fluids find use in sterilizing medical equipment due to their ability to destroy the cell walls of some kinds of yeast, bacteria, viruses, and fungi. However, this property does not see widespread use as the mechanism has yet to be fully understood.
Impregnation
Impregnation processes can be carried out via supercritical fluids. Supercritical fluids like CO2 dissolve the active substance and deposit onto the solid surface when the mixture is passed over. An important application is the dyeing of polymer fibers such as polyester.
Drying
The lack of surface tension in supercritical fluids allows them to dissolve solvents from highly porous structures without changing the internal 3D structures, thus leaving a dried solvent-free material with an undamaged internal system. This technique finds use in dry-cleaning services to replace undesirable chlorinated solvents.