Atomic Radius
What is Atomic Radius
Atomic radius is the distance of the outermost or valence electrons from the nucleus of an atom. Suppose one visualizes the atom to be a sphere where the nucleus lies at the center, and the outermost electrons lie at the boundary. Then, the radius of the sphere represents the atomic radius [1-4].
How to Find Atomic Radius From Chemical Bonds
There is no equation or formula for atomic radius. According to Heisenberg Uncertainty Principle, the electrons do not remain fixed in their orbits. Hence, their positions cannot be determined accurately. It is for this reason that the atomic radius is challenging to measure. In order to get an estimate of the atomic radius, scientists generally determine it from the distance between the nuclei of two atoms bonded in a chemical bond. This distance is typically measured in picometer (pm).
Based on this concept, the atom will have different radii depending on the type of the chemical bond [1].
1. Covalent Radius
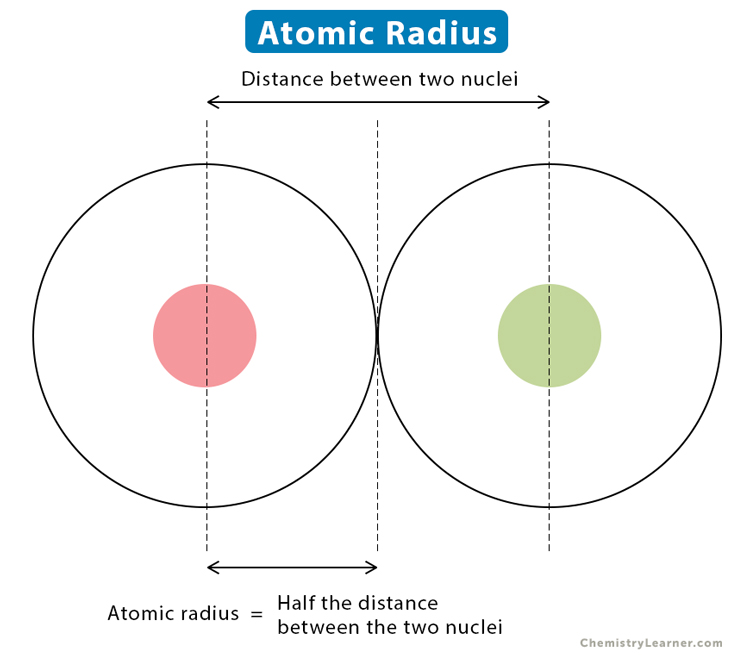
The covalent radius is a measure of the atomic size when it is bonded with another atom in a covalent bond. When two identical atoms are bonded together, the covalent radius is exactly one-half the distance between the two nuclei.
Example: In a hydrogen molecule (H2), the interatomic distance between the two hydrogen atoms is 74 pm. Hence, the covalent radius of the hydrogen atom is 37 pm.
2. Ionic Radius
The ionic radius is defined as the radius of an ion in an ionic bond. It is determined from the distance between two ions and dividing it according to the atomic sizes.
Example: The ionic radius of lithium ion (Li+) is 76 pm.
3. Metallic Radius
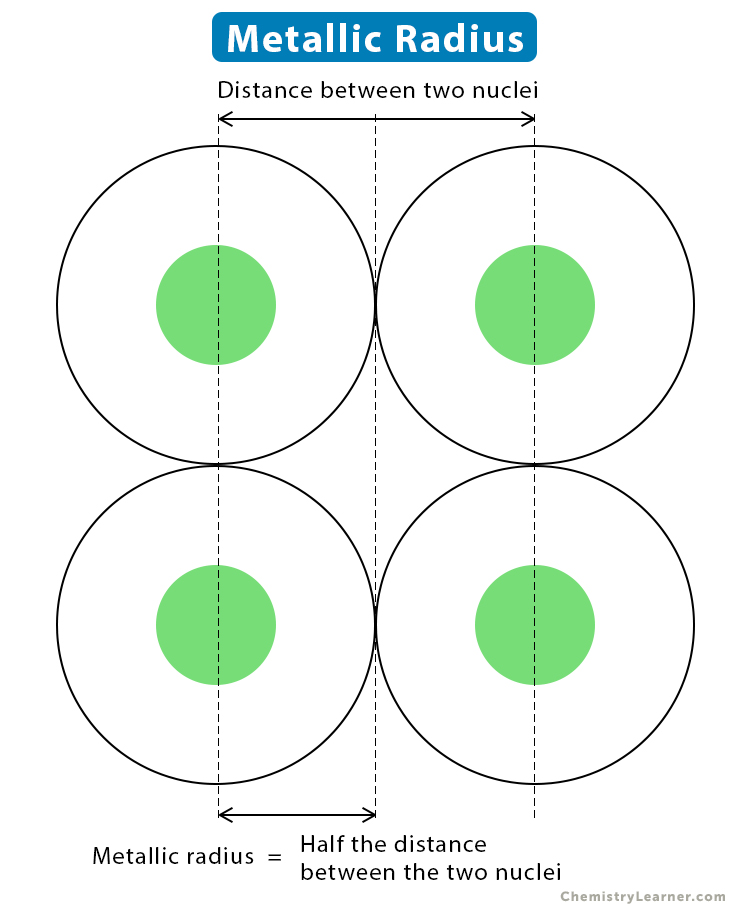
The metallic radius is defined as one-half of the distance between the nuclei of two adjacent atoms in a metallic bond. Since metal atoms are present in a cluster, the distances between any two nuclei are the same for all pairs of atoms.
Example: The metallic radius of sodium (Na) is 190 pm.
4. Van der Waals Radius
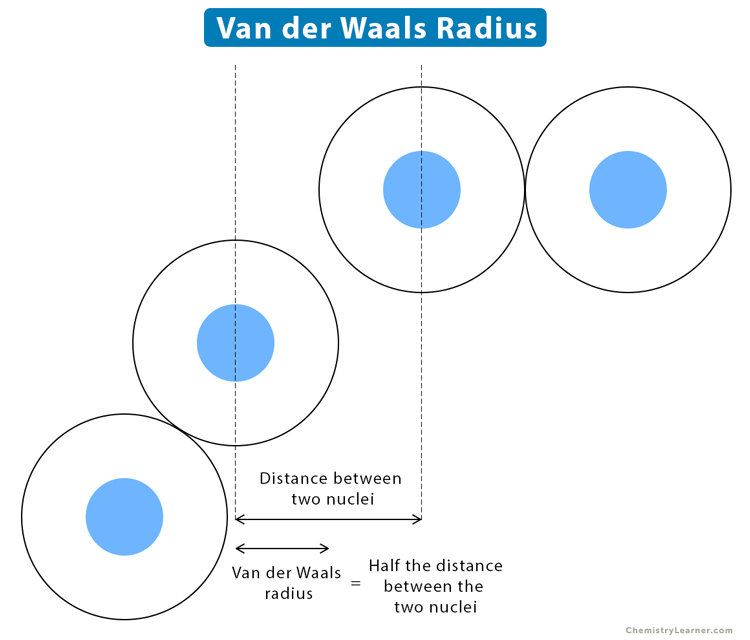
The van der Waals radius is equal to one-half the distance between two unbonded atoms held by weak but balanced electrostatic force, known as van der Waals forces.
Example: The van der Waals radius of oxygen (O) is 152 pm.
Each element of the periodic table has an atomic radius, which can be determined from the bond it forms and the atomic number. Hence, each element can have more than one radius.
Atomic Radius Periodic Trend
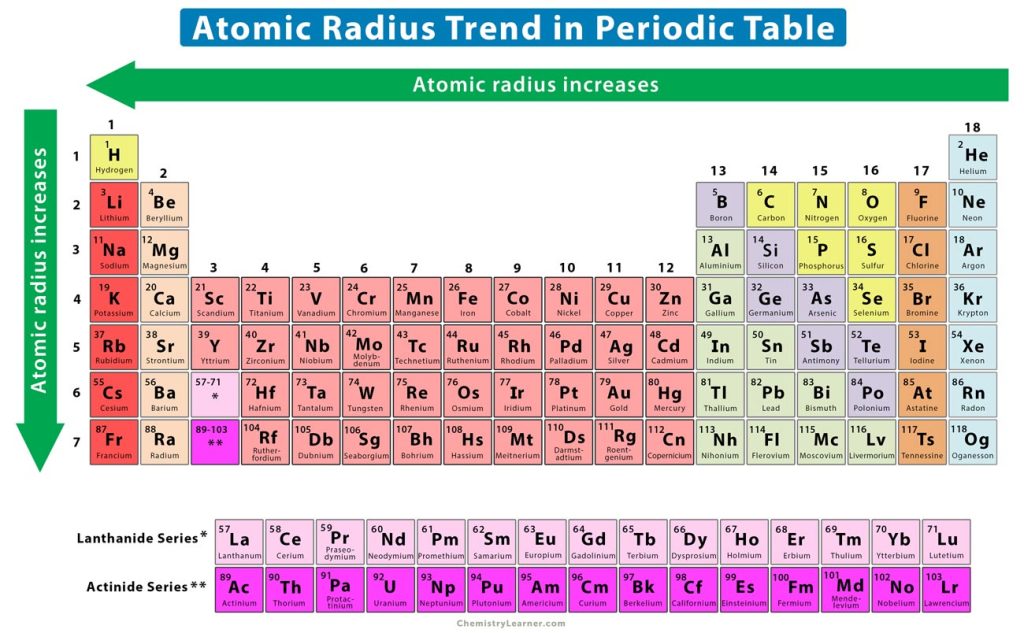
A periodic trend is a specific pattern of an atom’s property observed among the elements as one moves from left to right across a period and top to bottom down a group of the periodic table. The atomic radius displays a specific trend. All the atomic radii discussed above show a similar trend. Values of the atomic radii of a few elements are given in the chart at the end of this section [1-4, 6].
1. Horizontal Trend: Atomic Radius Across a Period
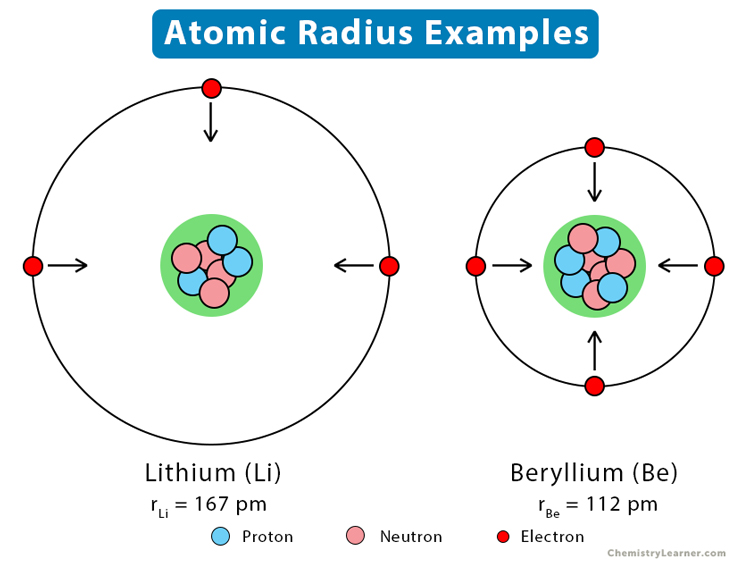
From left to right along a period, the atomic number of the elements gradually increases. Electrons are added to the same shell of an atom. Protons are also added to the atomic nucleus, making the nucleus more positively charged. As a result, the electrostatic attraction between the electrons and nucleus increases, and the outermost or valence electrons are held closer to the nucleus. The following image shows the atomic radii of lithium and beryllium, which lie in groups 1 and 2 respectively of the second period.
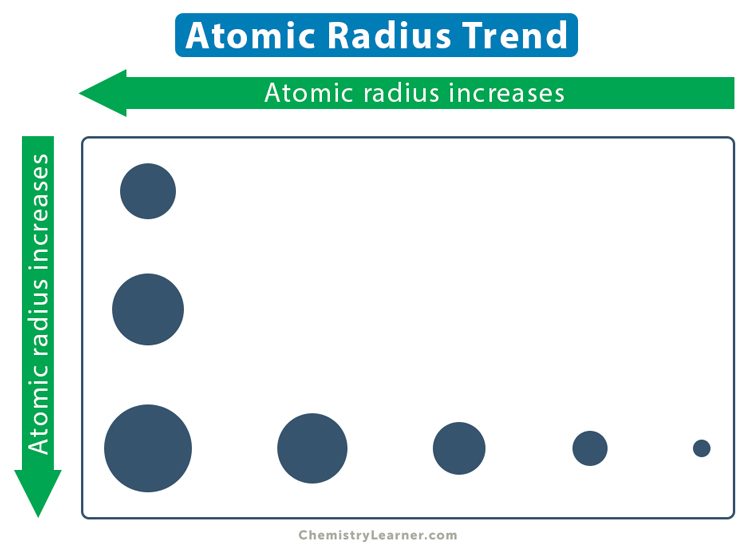
Thus, the atomic radius gradually decreases from left to right of a period. In other words, the atomic radius increases from right to left, as shown in the trend image above.
2. Vertical Trend: Atomic Radius Down a Group
From top to bottom down a group, the atomic number dramatically increases. It is evident that as the atomic number increases, the valence electrons occupy higher shells. The inner electrons shield the valence electrons and reduce the attractive force between the valence electrons and nucleus. As a result, the valence electrons remain further away from the nucleus. Therefore, the atomic size and atomic radius increase from top to bottom.
In order to show the variation across the periods and down the groups, the atomic radius values of the elements in the periodic table are shown in the chart below [1].