Helium
Table of Contents
What is Helium
Helium (pronunciation: HEE-lee-em) is a colorless, odorless element classified as a non-metal, belonging to the group of inert gases in the periodic table and is represented by the chemical symbol He [1, 2, 3]. It is a monoatomic gas that forms weak diatomic molecules only at temperatures close to -273.15 °C or “the absolute zero” [4].
Isotopes
Helium has nine known isotopes out of which 3He and 4He are stable with natural abundances of about 0.0002% and 99.9998% respectively [5]. Its radioactive isotopes are short-lived, with 6He being the longest-lived characterized by a half-life period of 806.7 milliseconds while 5He is the least stable with a half-life period of 7.6 X 10-22 seconds [5].
Where is Helium Found
Helium is the second-most abundant element found in the universe only after hydrogen [1]. It is found in all stars and is formed on the Earth from radioactive elements when they undergo alpha-decay [1]. For natural dynamic balance, the low-density helium escapes into the Earth’s atmosphere and then to outer space [1]. The price of extracting the element from the air is very high, and so, it is commercially obtained from natural gas that can contain about 7% helium by volume [1].
The top 3 helium producers are USA, Algeria, and Russia while top helium reserve holding countries include USA, Qatar, and Algeria [1].
History
Origin of its Name: It is derived from ‘Helios’, Greek for the Sun, as it was first identified as the gas that constitutes the Sun’s corona [1]
Who discovered it: The element was discovered by the Scottish chemist Sir William Ramsay and independently by the Swedish chemists Nils Abraham Langlet and Per Teodor Cleve [1, 2].

Liquid Helium
When and How was it Discovered
Before the discovery of helium on Earth, it was found on the Sun [2]. In 1868, the French astronomer Pierre Janssen studied a total solar eclipse in India and found a yellow line while measuring the Sun’s spectrum [1, 2]. An English astronomer, Sir Joseph Norman Lockyer, also observed the same spectral line and presumed that it was caused by an unknown metallic element, which he named helium [1, 2].
In 1895, William Ramsay experimented with cleveite, a radioactive mineral containing uranium and collected the gas that formed after its dissolution in sulfuric acid [1, 2]. He sent the sample to Sir William Crookes and Norman Lockyer, who identified the helium within it [2]. In 1895, at Uppsala, Sweden, Abraham Langlet and Teodor Cleve independently collected the gas produced after dissolving uraninite (UO2) in acid [1, 2]. They found its atomic weight and declared that it was helium [1].
Helium Identification |
|||
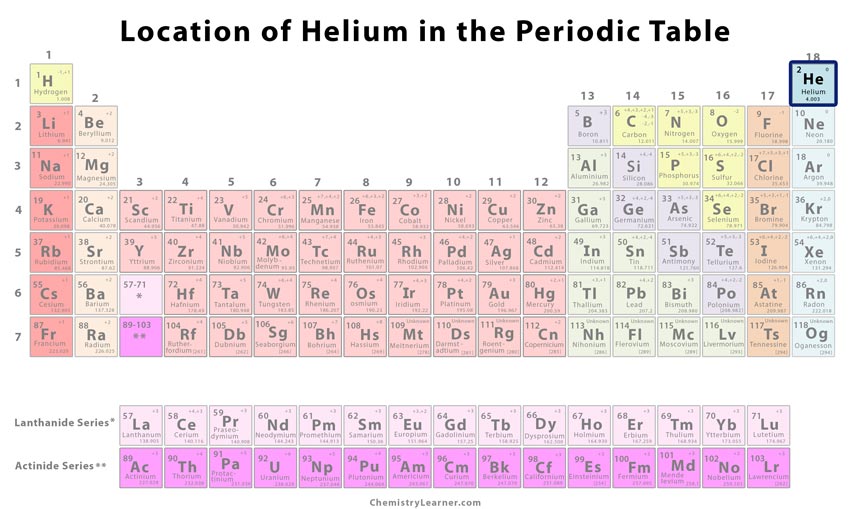
| Atomic number | 2 [1] | ||
| CAS number | 7440-59-7 [1] | ||
| Position in the periodic table | Group | Period | Block |
| 18 [1] | 1 [1] | s [1] | |
Properties and Characteristics of Helium
General Properties |
||||||||||||||||
| Relative atomic mass | 4.003 [1] | |||||||||||||||
| Atomic mass | 4.003 amu [1] | |||||||||||||||
| Molar mass | 4.0026020 ± 0.0000020 g/mol [6] | |||||||||||||||
| Molecular weight | 4.003 g/mol [7] | |||||||||||||||
| Mass number | 4 [8] | |||||||||||||||
Physical Properties |
||||||||||||||||
| Color | Colorless [1, 4] | |||||||||||||||
| Melting point/freezing point | Unknown [1] | |||||||||||||||
| Boiling point | -268.928 °C, -452.07 °F [1] | |||||||||||||||
| Density | 0.000164 g cm-3 [1] | |||||||||||||||
| State of matter at room temperature (solid/liquid/gas) | Gas [1, 4] | |||||||||||||||
| Thermal (heat) conductivity | 0.1513 W/(m K) [9] | |||||||||||||||
| Specific heat | 5193 J kg-1 K-1 [1] | |||||||||||||||
| Bulk modulus | Unknown [1] | |||||||||||||||
| Shear modulus | Unknown [1] | |||||||||||||||
| Young’s modulus | Unknown [1] | |||||||||||||||
| Vapor pressure | ||||||||||||||||
| – Temperature (K) | 400 | 600 | 800 | 1000 | 1200 | 1400 | 1600 | 1800 | 2000 | 2200 | 2400 | |||||
| – Pressure (Pa) | – | – | – | – | – | – | – | – | – | – | – | |||||
Chemical Properties |
||||||||||||||||
| Oxidation state/Oxidation number | 0 [10] | |||||||||||||||
| Isotopes | Isotope | Mass | Abundance (%) | Half-life | Mode of decay | |||||||||||
| 3He | 3.016 | 0.000134 | – | – | ||||||||||||
| 4He | 4.003 | 99.9999 | – | – | ||||||||||||
Atomic Data of Helium (Element 2)
| Valence electrons | 2 [11] | |||||||
| Quantum numbers | ||||||||
| – n | 1 [11] | |||||||
| – ℓ | 0 [11] | |||||||
| – mℓ | 0 [11] | |||||||
| – ms | -1/2 [11] | |||||||
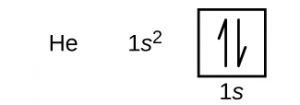
| Electron configuration (noble gas configuration) | 1s2 [1] | |||||||
| Atomic structure | ||||||||
| – Number of electrons | 2 [4, 8] | |||||||
| – Number of neutrons | 2 [4, 8] | |||||||
| – Number of protons | 2 [4, 8] | |||||||
| Radius of Atom | ||||||||
| – Atomic radius | 1.400 Å [1] | |||||||
| – Covalent radius | 0.37 Å [1] | |||||||
| Electronegativity (Pauling-scale) | Unknown [1] | |||||||
| Electron affinity | Not stable [1] | |||||||
| Ionization energy (kJ mol-1) | 1st | 2nd | 3rd | 4th | 5th | 6th | 7th | 8th |
| 2372.322 | 5250.516 | – | – | – | – | – | – | |
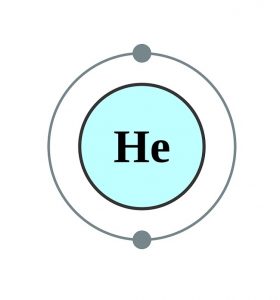
Helium Electron Configuration (Bohr Model)
What is Helium Used for
- As a coolant, helium is used in MRI scanners’ superconducting magnets, NMR spectrometers, and the Large Hadron Collider [1]. It is used to cool instruments in satellites and space vehicles [1].
- The gas is used to fill weather balloons, airships, and decorative blimps due to its low density [1, 4]. Despite hydrogen being more buoyant and cheaper, helium is preferred because it is non-flammable [15].
- It can be used for pressurizing and cooling the liquid fuel (liquid hydrogen) in rockets and space vehicles [1, 4].
- Since helium is extremely unreactive by nature, it can provide a protective atmosphere for arc welding, as well as manufacturing semiconductors and fiber optics [1, 4].
- For people working under pressurized conditions, like deep-sea divers, 80% helium is mixed with 20% oxygen to create an artificial atmosphere [1, 4]. Divers use helium because it leaves their body faster as compared to nitrogen, helping in faster decompression [4].
- It is used for detecting leaks in air-conditioning systems of automobiles and for inflating car airbags after the accident [1].
- Helium-neon laser barcode scanners are used in supermarket checkouts [1].
- Electron microscopes are now replaced by helium-ion microscopes for their better image resolution [1].
Helium Toxicity and Health Effects
Helium is not known to be toxic and does not play any biological role [1]. Although it is commonly believed that inhaling it causes the voice to temporarily become high-pitched [4, 12], heavily inhaling helium can cause an obstruction, cutting off oxygen supply [13].
Interesting Facts
- The graphical representation of helium shows the Sun, indicating the origin of its name from the Greek word ‘Helios’ and its presence in the Sun [1].
- The Federal Helium Reserve near Amarillo, Texas has about 40% of the helium gas used by the Americans in a year [3].
- Since helium turns into a superfluid state at about -271 °C, it is used by the scientists for studying the behavior of superfluids [3].
Cost of Helium
While the price of pure helium is about $0.052 per gram, liquid helium costs roughly $5 per liter [4, 14].
- References
- http://www.rsc.org/periodic-table/element/2/helium
- https://education.jlab.org/itselemental/ele002.html
- https://www.livescience.com/28552-facts-about-helium.html
- https://www.chemicool.com/elements/helium.html
- https://education.jlab.org/itselemental/iso002.html
- https://www.webqc.org/molecular-weight-of-helium.html
- https://pubchem.ncbi.nlm.nih.gov/compound/helium
- https://hobart.k12.in.us/ksms/PeriodicTable/helium.htm
- http://periodictable.com/Elements/002/data.html
- https://www.easycalculation.com/chemistry/oxidation-number-helium.html
- http://chemistry-reference.com/q_elements.asp?Symbol=He&language=en
- https://www.medscape.com/viewarticle/760299
- https://www.medscape.com/viewarticle/760299
- https://physicstoday.scitation.org/doi/pdf/10.1063/PT.3.3424
- https://www.independent.co.uk/news/science/why-the-world-is-running-out-of-helium-2059357.html