Aluminum Nitride
Aluminum nitride, referred to as azanylidynealumane in IUPAC nomenclature, is an inorganic compound of aluminum and nitrogen represented by the chemical formula AlN [1]. It is not a naturally-occurring compound and has to be artificially synthesized [2]. It is characterized by high thermal conductivity and electrical resistivity, as well as an excellent corrosion resistance [2].
How is Aluminum Nitride Prepared
Its synthesis involves carbothermal reduction of aluminum oxide (Al2O3) using gaseous nitrogen at a temperature of 1400-1700 °C [6]:
- Al2O3 + C → Al2O2 + CO
- Al2O2 + 2C + N2 → 2AlN + 2CO
Sintering aids like CaO or Y2O3 are used, and then the mix is hot pressed to produce the dense technical-grade material.
Properties and Characteristics of Aluminum Nitride
General Properties |
|
| Molar Mass/Molecular Weight | 40.989 g/mol [1] |
Physical Properties |
|
| Color and Appearance | White to pale yellow powder or sheet [3, 4] |
| Melting Point | 2200 °C, 3990 °F [3] |
| Boiling Point | 2517 °C, 4563 °F [3] |
| Density | 2.9-3.3 g cm-3 [3] |
| State of matter at room temperature (solid/liquid/gas) | Solid [3] |
| Solubility | Slightly soluble in mineral acids, decompose in water solutions containing acids and bases [7] |
| Solubility in Water | Insoluble (monocrystalline form), hydrolyzes (powder form) |
| Thermal Conductivity | 80-200 W/K.m [3] |
| Volume Resistivity | 1010-1016 Ω.cm [8] |
| Dielectric Constant | 9.14 [9] |
| Hardness | 1100 Kg/mm2 [10] |
| Thermal Emissivity (100 °C) | 0.93 [11] |
| Infrared Refractive Index (300 K) | 2.1-2.2 (monocrystals and epitaxial films), 1.9-2.1 (polycrystalline films), 1.8-1.9 (amorphous films) [9] |
| Lattice Constant (300 K) a and c | 3.11(1) Å and 4.98(1) Å [9] |
Atomic Properties |
|
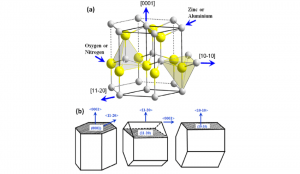
| Crystal Structure | Wurtzite |
What is It Used for
- A thin film of crystalline aluminum nitride, because of its piezoelectric properties, is used for developing small surface acoustic wave sensors (SAWs) on silicon wafers [12].
- RF filters in mobile phones consist of a microelectromechanical system (MEMS) device, called thin film bar acoustic resonator (FBAR), which uses AlN [13].
- A piezoelectric layer of AlN is used in making micromachined ultrasound transducers (MUTs) that have applications in velocity sensing, robotics, and medical imaging [14].
- Aluminum nitride nanotubes are used as chemical sensors for detecting toxic gases [15].
- Currently, there is research into manufacturing light-emitting diodes that use gallium-aluminum nitride-based semiconductors for improved emission efficiency [16].
Aluminum Nitride Toxicity
Exposure to AlN through mouth, inhalation, or injection may cause bone and lung toxicity [17]. Repeated exposure can irritate the eyes and skin [17].
- References
- Aluminum Nitride (AlN) – Pubchem.ncbi.nlm.nih.gov
- Aluminum Nitride (AlN) – Surmet.com
- Aluminum Nitride – Americanelements.com
- Aluminium nitride – Chemspider.com
- CHEBI:50884 – aluminium nitride – Ebi.ac.uk
- Carbothermal Synthesis Of Aluminum Nitride Using Sucrose – Digitool.library.mcgill.ca
- Aluminum nitride – Chemicalbook.com
- Effects of adding Y2O3 on the electrical resistivity of aluminum nitride ceramics – Jstage.jst.go.jp
- AlN – Aluminum Nitride Basic Parameters – Ioffe.ru
- Aluminum Nitride, AlN Ceramic Properties – Accuratus.com
- Aluminum Nitride Property List – Matsuo-sangyo.co.jp
- Aluminum Nitride on Sapphire Films for Surface Acoustic Wave Chemical Sensors – Jhuapl.edu
- AlN and ScAlN Contour Mode Resonators for RF Filters – Osti.gov
- Aluminum Nitride Piezoelectric Micromachined Ultrasound Transducer Arrays – Semanticscholar.org
- Aluminum Nitride Nanotubes – Researchgate.net
- Aluminum nitride nanowire light emitting diodes: Breaking the fundamental bottleneck of deep ultraviolet light sources – Nature.com
- Aluminum Nitride Safety Data Sheet – Espimetals.com








thank you so mch