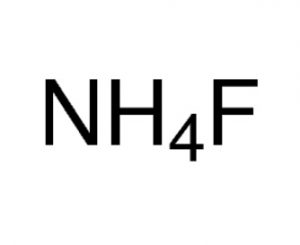
Ammonium Fluoride
Ammonium fluoride is a crystalline, noncombustible inorganic compound represented by the chemical formula NH4F or FH4N [1, 2]. It is a fluoride salt with ammonium as the counterion and is corrosive to cement, glass, and most metals, including aluminum [1, 2].
How is Ammonium Fluoride Prepared
It is synthesized by the addition of ammonia gas to cold hydrofluoric acid at a temperature of about 10 °C represented by the reaction [4]:
NH3 + HF → NH4F
It can be prepared by using silicon tetrafluoride gas (SiF4) and high purity ammonia (NH3) as reactants, which is given in the following equations [4]:
3SiF4 + 2H2O → 2H2SiF6 + SiO2
H2SiF6 + 2NH3 → (NH4)2SiF6
(NH4)2SiF6 + 4NH3 + 2H2O → SiO2 + 6NH4F
Reactions with Other Compounds
Ammonium Fluoride and Water
When NH4F reacts with water, it produces hydrofluoric acid and ammonium hydroxide, as represented by the following equation [2]:
NH4F + H2O → NH4OH + HF
Calcium Nitrate and Ammonium Fluoride
Ammonium fluoride reacts with calcium nitrate to form calcium fluoride, dinitrogen oxide, and water:
Ca(NO3)2 + 2NH4F → CaF2 + 2N2O + 4H2O
Ammonium Fluoride and Iodine
When ammonium fluoride and iodine are used as reactants, they undergo a single displacement reaction to form ammonium iodide and fluorine:
NH4F + I2 → NH4I2 + F
Properties and Characteristics of Ammonium Fluoride
General Properties |
|
| Molar Mass/Molecular Weight | 37.037 g/mol [1] |
Physical Properties |
|
| Color and Appearance | White or colorless crystalline powder [1] |
| Melting Point | 238 °C, 460.4 °F [1] |
| Boiling Point | N/A |
| Density | 1.01 g cm-3, 1.32 at 77 °F [1] |
| pH | Aqueous NH4F is acidic [1] |
| State of matter at room temperature | Dry powder or liquid [1] |
| Solubility | Insoluble in liquid NH3, slightly soluble in alcohol [1] |
| Solubility in Water | 83.5 g/100 g at 25 °C [1] |
| Magnetic Susceptibility (χ) | -23.0 X 10-6 cm3/mol |
Atomic Properties |
|
| Crystal Structure | Hexagonal wurtzite |
Uses
- For etching glass, polishing metal surfaces, extracting rare metals, and disinfection and preservation in the brewing industry [5]
- As masking agents, preservatives, and reagents for analysis [5]
- For wood preservation, complexometric titration of aluminum, spot analysis of zirconium, and glass engraving [5]
Is It Safe
NH4F causes acute toxicity when it is swallowed, inhaled, or comes in contact with skin [1]. Its inhalation may result in irritation of the respiratory tract and ingestion of small quantities of soluble fluorides can also be fatal [1]. Its contact with skin causes burns, and exposure through eye contact can irritate the mucous membrane [1].
- References
- Ammonium Fluoride – Pubchem.ncbi.nlm.nih.gov
- Ammonium Fluoride – Cameochemicals.noaa.gov
- Ammonium Fluoride – Chemspider.com
- Synthesis of Ammonium Fluoride: A Literature Review – Repository.up.ac.za
- Ammonium Fluoride – Chemicalbook.com