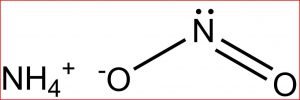
Ammonium Nitrite
Ammonium nitrite represented by the chemical formula NH4NO2 or H4N2O2 [1] that bears the IUPAC name azanium nitrite [4] is a compound containing nitrogen, hydrogen and oxygen that is soluble in water [6]. It is an ammonium salt and an ionic compound [1].
Ammonium Nitrite Identification |
|
| CAS Number | 13446-48-5 [1] |
| PubChem CID | 26004 [1] |
| ChemSpider ID | 24223 [2] |
| EC Number | 236-598-7 [1] |
How to Make it
Ammonium nitrite can be prepared by a reaction between silver nitrite and ammonium chloride that also produces silver chloride [6].
2AgNO2 + NH4Cl = NH4NO2 + 2AgCl
Nitrous acid also reacts with ammonium hydroxide to produce ammonium nitrite and water [7].
HNO2 + NH4OH = NH4NO2 + H2O
Properties and Characteristics of Ammonium Nitrite
General Properties |
|
| Molar mass/molecular weight | 64.044 g/mol [1] |
Physical Properties |
|
| Color/appearance | White-yellowish crystals [5] |
| Odor | Odorless [6] |
| Melting point/freezing point | Decomposes (explodes at 60°C-70°C) [3] |
| Boiling point | N/A [4] |
| Density | 1.69 g cm-3 [3] |
| State of matter at room temperature (normal phase) | Solid [5] |
Chemical Properties |
|
| Solubility in water | 64.3 g/100g (at 19.15oC) [5] |
| pH | >7 (basic) [6] |
Prominent Reactions
If the concentrated solution of the compound is heated too strongly, it explodes decomposing into nitrogen gas and water [5, 6].
NH4NO2 = N2 + 2H2O
Ammonium Nitrite Uses
- As a microbiocide, rodenticide and agricultural pesticide [4].
- For the synthesis of nitrogen gas and ammonium cobalt-nitrite [6].
- In making explosives [6].
Is It Dangerous
The compound is highly toxic to humans and aquatic animals. It can cause an explosion hazard. Heating it can produce toxic fumes of ammonia and nitrogen oxides [4, 6]
Ammonium Nitrite (NH4NO2) Cost
The unstable compound is extremely difficult to find in its pure form and almost none of the major chemical suppliers sell it for commercial purposes [6].
- References
- Ammonium Nitrite – Pubchem.ncbi.nlm.nih.gov
- Ammonium Nitrite – Chemspider.com
- Ammonium Nitrite Formula – Byjus.com
- Ammonium Nitrite – Lookchem.com
- Ammonium Nitrite, 20% Soln – Chemicalbook.com
- Ammonium Nitrite – http://www.sciencemadness.org/smwiki/index.php/Ammonium_nitrite
- Nitrous acid react with ammonium hydroxide – Chemiday.com