Beryllium Hydroxide
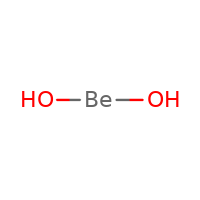
Beryllium hydroxide, also known as hydrated beryllia and beryllium dihydroxide, is a metal hydroxide that has amphoteric properties (behaves both as an acid and base) and chemically represented by Be(OH)2 [1, 2]. The pure beryllium hydroxide occurs naturally as the rare, orthorhombic mineral behoite, and very rare monoclinic clinobehoite [3].
How is Beryllium Hydroxide Prepared
Pure beryllium hydroxide is synthesized as a by-product during the extraction of beryllium from mineral ore beryl (Be3Al2Si6O18) or bertrandite (Be4Si2H2O9) [5]. The ore is melted, solidified, crushed, and then treated with sulfuric acid to produce a water-soluble sulfate [5]. The sulfate undergoes several chemical extraction procedures so that all contaminants are removed and beryllium hydroxide is formed as a precipitate [5].
Reactions with Other Compounds
Beryllium Hydroxide and Sodium Hydroxide
When a concentrated solution of sodium hydroxide reacts with beryllium hydroxide, it dissolves to produce sodium tetrahydroxoberyllate [2, 6]:
Be(OH)2 + 2NaOH → Na2[Be(OH)4]
Beryllium Hydroxide and Hydrochloric Acid
It reacts with dilute hydrochloric acid to form beryllium chloride, as indicated by the following chemical equation [2]:
Be(OH)2 + 2HCl → BeCl2 + 2H2O
Properties and Characteristics of Beryllium Hydroxide
General Properties |
|
| Molar Mass/Molecular Weight | 43.026 g/mol [1] |
Physical Properties |
|
| Color and Appearance | White, opaque crystalline powder [7] |
| Density | 1.92 g cm-3 [7] |
| Melting Point | 138 ° C, 280.4 °F (decomposes) [7] |
| Boiling Point | N/A [7] |
| State of matter at room temperature | Solid |
| Solubility | Soluble in hot sodium hydroxide and acids, slightly soluble in dilute alkali [8] |
| Solubility in Water | Almost insoluble in water (0.0000023965 g per L) [8] |
| Heat Capacity (C) | 1.443 J K-1 |
Atomic Properties |
|
| Crystal Structure | Linear with tetrahedral beryllium centers |
What Is It Used for
- As a source in the manufacture of beryllium and its oxide [8].
- In braking systems, engines, altimeters, and precision tools in the aerospace industry [9].
- Producing anti-lock brake systems, steering wheel components, and air-bag sensors [9].
- Manufacturing medical laser components, X-ray tube windows, and dental crowns [9].
- Making nuclear reactor components, missile guidance systems, and heat shields in defense industry [9].
Is It Safe
Beryllium hydroxide is known to be carcinogenic and repeated exposure through contact, inhalation, or ingestion may cause organ toxicity [1]. It is also hazardous to aquatic life [1].
- References
- Beryllium Hydroxide – Pubchem.ncbi.nim.nih.gov
- Some Beryllium Chemistry Untypical Of Group 2 – Chemguide.co.uk
- Beryllium Dihydroxide (13327-32-7) – Chemchart.com
- Beryllium Hydroxide – Chemspider.com
- Sources of Beryllium – Berylium.com
- Beryllium Hydroxide React with Sodium Hydroxide – Chemiday.com
- Beryllium Hydroxide – Americanelements.com
- Beryllium Hydroxide – Chemicalbook.com
- Beryllium Hydroxide Market Analysis By Application (Aerospace, Automotive, Biomedical, Defence, Electrical and telecommunications), By Region (North America, Europe, Asia Pacific, Latin America And Middle East & Africa), 2014 To 2025 – Grandviewresearch.com






can the beryllium hydroxide be dissolved in aqueous ammonia or ammonia water or ammonium hydroxide
It is moderately soluble in aqueous ammonia or ammonia water and ammonium hydroxide.