Calcium Nitrate
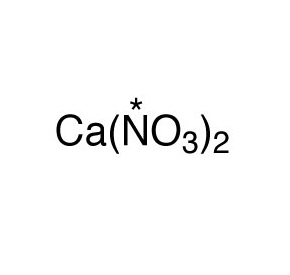
Calcium Nitrate, commonly called lime nitrate or Norwegian saltpeter, is a simple inorganic salt of calcium, nitrogen, and oxygen represented by the chemical formula Ca(NO3)2 or CaN2O6 [1, 2]. It occurs either as an anhydrous compound that can absorb moisture from the atmosphere or as a tetrahydrate with water molecules [3]. In IUPAC nomenclature, it is called calcium dinitrate [3].
Calcium Nitrate Composition and Synthesis
Calcium nitrate is produced by reacting calcium carbonate (normally as limestone) with nitric acid [4]:
CaCO3 + 2HNO3 → Ca(NO3)2 + CO2 + H2O
It is also obtained as an intermediate product during the production of nitrogen fertilizers through the Odda process, which involves acidification of calcium phosphate [4]:
Ca3(PO4)2 + 6HNO3 + 12H2O → 2H3PO4 + 3Ca(NO3)2 + 12H2O
It is synthesized through the reaction of calcium hydroxide and ammonium nitrate solution [4]:
2NH4NO3 + Ca(OH)2 → Ca(NO3)2 + 2NH4OH
Reaction with Other Compounds
Aqueous Calcium Nitrate and Aqueous Sodium Phosphate
When an aqueous solution of calcium nitrate is mixed with aqueous sodium phosphate, it produces calcium phosphate and sodium nitrate as represented by the following equation:
3Ca(NO3)2 + 2Na3PO4 → Ca3(PO4)2 + 6NaNO3
Calcium Nitrate and Ammonium Phosphate
Calcium nitrate reacts with ammonium phosphate to yield ammonium nitrate and calcium phosphate, which is shown in the following equation:
3Ca(NO3)2 + 2(NH4)3PO3 → 6NH4NO3 + Ca3(PO3)2
Calcium Nitrate and Sodium Acetate
When calcium nitrate reacts with sodium acetate, it undergoes double displacement reaction to form sodium nitrate and butanedioic acid, calcium salt:
Ca(NO3)2 + 2C2H3NaO2 → 2NaNO3 + Ca(C4H6O4)
Calcium Nitrate and Ammonium Fluoride
Calcium nitrate reacts with ammonium fluoride to form nitrous oxide, calcium fluoride, and water:
Ca(NO3)2 + 2NH4F → 2N2O + CaF2 + 4H2O
Calcium Nitrate and Magnesium Sulfate
Calcium nitrate undergoes a double displacement reaction with magnesium sulfate to yield calcium sulfate and magnesium nitrate:
Ca(NO3)2 + MgSO4 → CaSO4 + Mg(NO3)2
Calcium Nitrate and Potassium Chloride
When mixed with potassium chloride, it produces calcium chloride and potassium nitrate as given by the following equation:
Ca(NO3)2 + 2KCl → CaCl2 + 2KNO3
Calcium Nitrate and Sodium Carbonate
They undergo a double displacement reaction and form sodium nitrate and calcite as the products:
Ca(NO3)2 + Na2CO3 → 2NaNO3 + CaCO3
Calcium Nitrate and Sodium Chloride
Calcium nitrate reacts with halite or rock salt to yield calcium chloride and sodium nitrate:
Ca(NO3)2 + 2NaCl → CaCl2 + 2NaNO3
Calcium Nitrate and Sodium Hydroxide
When aqueous calcium nitrate reacts with aqueous sodium hydroxide, it forms an insoluble solid calcium hydroxide and an aqueous solution of sodium nitrate:
Ca(NO3)2 + 2NaOH → Ca(OH)2 + 2NaNO3
Calcium Nitrate and Water
Ca(NO3)2 dissociates in H2O to form Ca2+ and NO3- ions:
Ca(NO3)2 ↔ Ca2+ + 2 NO3-
Properties and Characteristics of Calcium Nitrate
General Properties |
|
| Molar Mass/Molecular Weight | 164.086 g/mol (anhydrous) [3] |
Physical Properties |
|
| Color and Appearance | Colorless to white or light gray granular powder, hygroscopic crystals [3] |
| Melting Point | About 560 °C, 1042 °F (anhydrous) [3] |
| Boiling Point | Decomposes (anhydrous); 132 °C, 270 °F (tetrahydrate) [5] |
| Density | 2.5 g cm-3 [3, 5] |
| pH | 5-7 [7] |
| State of matter at room temperature | Solid [3] |
| Solubility | Soluble in methanol, acetone, and anhydrous ammonia, almost insoluble in nitric acid [3] |
| Solubility in Water | 121.2 g/L [3] |
| Magnetic Susceptibility (χ) | -45.9 X 10-6 cm3/mol |
Atomic Properties |
|
| Crystal Structure | Cubic (anhydrous), monoclinic (tetrahydrate) |
Uses
- As fertilizer in agriculture, hydroponics, and greenhouse trades [3, 6].
- Treating wastewater and preventing odor emission [6].
- Applied with concrete and mortar in set-accelerating admixtures [6].
- In regenerable cold packs since the dissolution of calcium nitrate is extremely endothermic [6].
- As a constituent of molten salt mixtures that are used as heat-storage and heat-transfer fluid replacing thermo oil in solar power plants [6].
Is It Safe
Calcium nitrate causes acute toxicity and can be harmful if swallowed [3]. Its contact with skin or eyes also results in severe irritation or damage [3]. Calcium nitrate, being a strong oxidizer, may cause fire and give off toxic fumes [3].
- References
- Calcium Nitrate – Chemspider.com
- Calcium Nitrate – Americanelements.com
- Calcium Nitrate – Pubchem.ncbi.nlm.nih.gov
- Preparation of Calcium Nitrate – Rsc.org
- Calcium Nitrate – Americanelements.com
- Calcium Nitrate – Thechemco.com
- Calcium Nitrate – Ipni.net